Prof. J. Heathcote, Chair (IASL)
Prof. A. Elewaut (WGO)
Prof. S. Fedail (WGO)
Prof. A. Gangl (WGO)
Prof. S. Hamid (WGO)
Prof. M. Shah (WGO)
Dr. D. Lavanchy (WHO)
Dr. H. Liu (WGO)
Dr. J. Krabshuis (WGO)
1. Definition
Acute viral hepatitis (AVH) is a systemic infection predominantly affecting the liver. It is most often caused by viruses that are hepatotropic (hepatitis A, B, C, D, and E). Other viral infections may also occasionally affect the liver, such as cytomegalovirus (CMV), herpes simplex, coxsackievirus, and adenovirus. Whereas hepatitis A and E are self-limiting, infection with hepatitis C and to a lesser extent hepatitis B usually become chronic.
2. Introduction and key points
Possibly the key difference between this World Gastroenterology Organisation (WGO) guideline and other publications on acute viral hepatitis is an awareness of the potential for mismanagement. After all, the single most important issue in the management of acute viral hepatitis is that — in the great majority of cases —treatment should be supportive and does not require hospitalization or medication. Surgical intervention (during the acute state, or because of misdiagnosis) may bedangerous. Anecdotal evidence suggests there is often mismanagement of this condition, especially in poorly resourced settings.
Whereas most acute infections are asymptomatic, when symptoms are present they appear to be similar for all five types of virus. It is important to establish which virus is involved, as the risks of progression differ:
- Hepatitis A: self-limiting. The rate of fulminant hepatic failure (FHF) rate is very low; there is a 1% fatality rate in those over the age of 40.
- Hepatitis B: self-limiting in 95% of cases (adults only), but not in children under the age of 5.
- Hepatitis C: self-limiting in 20–50% of cases (>90% in those treated with interferon alpha monotherapy).
- Hepatitis D: self-limiting if HBV is self-limiting.
- Hepatitis E: self-limiting. The overall mortality rate in FHF is 1–3%; in pregnant women the rate is 15–25%.
For the most part, only supportive treatment is required. If HAV is endemic, hepatitis A infection can be excluded, as earlier infection induces lifelong immunity.
A second important issue is the identification of risk groups. In pregnant women, for example, it is very important to exclude HEV. Similarly, acute hepatitis is more dangerous in adults than in children, particularly in those over the age of 40, and moreso if there is a background of chronic liver disease. In all cases, risk groups for severe hepatitis should be identified; usually, this affects older adults and those with underlying chronic liver disease. Acute fulminant hepatitis may occur at any age.
Key points to remember:
- A case of acute “hepatitis” maybe caused by a virus, a toxin, or may be the first manifestation of a chronic liver disease.
- Acute viral hepatitis is almost always self-limiting.
- In almost every case, it is best to do nothing (except to stop medications such as the oral contraceptive pill).
- There is no role for vitamins; particularly in developing countries, intravenous vitamins are often given unnecessarily.
- There is no role for restriction of proteins in uncomplicated acute hepatitis. In developing countries, patients may already be deprived of
- proteins; this leads to protein deficiency, with the associated complications.
- Exercise should be started as soon as the patient feels fit to do so; there is no need to keep patients in bed.
- A raised serum alanine transaminase (ALT) level is the best indicator of acute hepatic injury, but does not reflect the severity of the disease; values for bilirubin and international normalized ratios (INR) are required for this.
- All forms of AHV show the same symptoms.
- The endemicity of the conditions should be taken into account — e.g., hepatitis A predominantly affects children in endemic areas.
- It should be ascertained whether the condition is an acute infection or a flare-up of a silent chronic infection.
- Other chronic liver diseases may present acutely — for example, autoimmune hepatitis.
- Consideration should be given to the transmission, and thus prevention and vaccination where appropriate.
- In a pregnant woman with HBV, the infant should be protected with hepatitis B vaccine and hepatitis B immunoglobulin (HBIg) at birth.
Fig. 1 Decision algorithm in the diagnosis of acute hepatitis
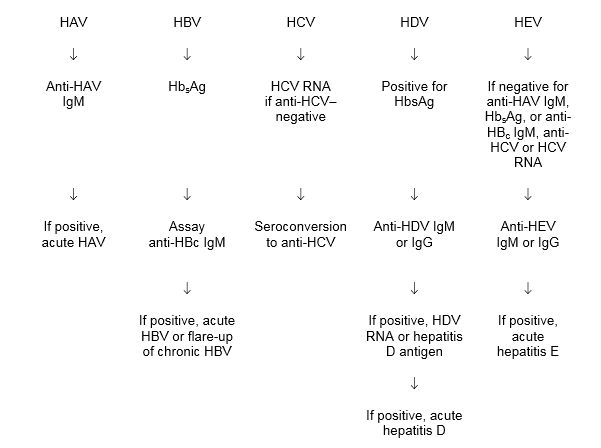
HAV, hepatitis A virus; HBc, hepatitis B core (antigen); HBsAg, hepatitis B surface antigen
HBV, hepatitis B virus; HCV, hepatitis C virus; HDV, hepatitis D virus; HEV, hepatitis E virus; IgG, immunoglobulin G; IgM, immunoglobulin M.
In acute HCV, it is important to test for HCV RNA and anti-HCV immediately.
- If both are present, the condition is probably a flare-up of chronic HCV.
- If only HCV RNA is present, then the condition is probably acute HCV; the patient should be followed up for the development of anti-HCV.
The onset of drowsiness and/or coagulopathy in a patient with acute viral hepatitis is always sinister.
Further information on AVH:
- Centers for Disease Control and Prevention (CDC): http://www.cdc.gov/
- Ryder SD, Beckingham IJ. ABC of diseases of liver, pancreas, and biliary system: acute hepatitis. BMJ 2001;322:151–3 (PMID: 11159575).
3. Acute hepatitis A
3.1 Pathogenesis and natural course
Hepatitis A virus (HAV) is an RNA-containing virus of the Picornaviridae family. The key feature is that it is a self-limiting disease. Management of HAV should therefore be supportive. The average HAV incubation time is 28 days, but it can vary from 15 to 45 days.
HAV infection is self-limiting and does not require treatment; contacts should be vaccinated (particularly in low incidence areas). There is no chronic infection; HAV infection induces lifelong immunity. Some people with HAV will have relapses after 6–9 months. The risk of fulminant hepatic failure is very low (0.01–0.1%), but increases with age and in those with preexisting liver disease. In patients over the age of 40, there is a 1% mortality rate.
3.2 Epidemiology: prevalence and incidence
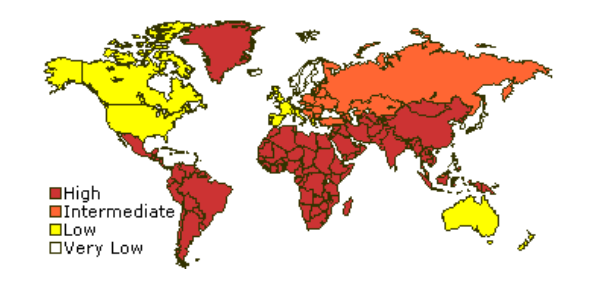
There are an estimated 1.5 million cases of acute hepatitis A annually worldwide, or — depending on local endemicity — around 150 per 100,000. Most of these occur in areas with poor hygiene and a poor sanitation infrastructure.
The prevalence and incidence of HAV infection are directly related to socioeconomic conditions. Infection occurs predominantly in childhood. Typically, more than 90% of children in developing countries are infected with HAV virus and have lifelong immunity.
Fig. 2 Prevalence of hepatitis A
3.3 Risk factors
Transmission. HAV is found in the feces of individuals with acute HAV in the presymptomatic and early phases of the disease. HAV is usually spread between individuals by oral contact with something that has been contaminated by fecal matter from an HAV-infected person — HAV transmission is fecal–oral. Most transmission is through contact with a household member, tourist travel to an endemic area, contact with a sex partner who has HAV, from an infection in an individual preparing food, or contact with babies in nurseries. Poor hygiene and poor sanitation pose the greatest risks.
Risk groups/circumstances for HAV:
- Children living in poor sanitation and in areas with low hygiene
- Children living in areas with a high incidence of HAV
- Those participating in anal sex
- Users of injected illegal drugs (homeless)
- Poor sanitation
- Consumers of high-risk foods (e.g., raw shellfish)
- Day-care employees and family of children in day care
- People traveling to endemic areas
3.4 Diagnosis and differential diagnosis
All forms of acute viral hepatitis have the same initial presentation. HAV virus is reliably diagnosed by anti-HAV immunoglobulin M (IgM); the presence of anti-HAV immunoglobulin G indicates a previous infection). Persistent anti-HAV IgM can sometimes be detected in patients with autoimmune hepatitis. In children, the illness is usually asymptomatic. In adults, HAV infection is usually symptomatic.
The principal symptoms are:
- Jaundice (yellowing of the skin and eyes)
- Fatigue
- Abdominal pain
- Loss of appetite
- Nausea
- Diarrhea
- Fever
- Dark urine
- Relapse with cholestasis or serum sickness
3.5 Management of acute hepatitis
The subjective impression of the patient should guide the doctor’s approach. Neither hospital admission, quarantine or bed rest, or medication (e.g., vitamin administration, dietary restrictions, blood transfusions) are necessary.
The treatment should be conservative and supportive. There is no specific medication for HAV infection. Hygiene is very important — hands should always be washed after bathroom use. The management should focus on treating the symptoms and identifying the small proportion of patients with a particular risk of developing fulminant hepatic failure. Patients over the age of 40 and those with underlying chronic liver disease are most at risk.
Contacts should be vaccinated.
Oral contraceptive treatment and hormone replacement therapy should be stopped to avoid cholestasis. Alcohol consumption is not advised.
3.6 Prevention
There are a number of inactivated vaccines on the market. Vaccination for preexposure prophylaxis (for example, with VAQTA or Havrix) provides long-term protection for up to 20 years. Vaccination for postexposure prophylaxis should be given as early as possible.
3.7 The future
Improving sanitation and water supplies are the most important goals in the prevention of HAV infection (as well as many other infections).
Key items are:
- Building a better public health infrastructure
- Improving sanitation and water supplies
- Establishing health visitor action programs (education and training, hygiene, and handwashing)
- Development of easy and low-cost diagnostic kits.
- Preexposure vaccination for those at risk (travelers, military personnel, male homosexuals, day-care employees).
- Postexposure vaccination for contacts of those with acute HAV.
- Always check for hepatitis B immunity and vaccinate when needed (the World Health Organization advocates universal vaccination for hepatitis B).
3.8 Further information
3.9 Summary
The clinical course of acute hepatitis A is indistinguishable from that of other types of acute viral hepatitis. However, if HAV IgM is positive, it is not necessary to check for other causes of acute viral hepatitis.
The symptoms typically include fever, malaise, anorexia, nausea and abdominal discomfort, and sometimes diarrhea, followed by dark urine and jaundice. The severity of the disease and the mortality increase in older age groups. Convalescence following hepatitis A may be slow, and is characterized by fatigue, nausea, and lack of appetite.
Complications of hepatitis A include relapsing hepatitis, with or without a cholestatic component, and fulminant hepatitis. Fulminant hepatitis occurs in approximately 0.01% of infections and is characterized by persistent nausea, vomiting, and bruising, with rapid deterioration in the level of consciousness and in liver function. There is a very high mortality rate.
Chronic infection with HAV does not occur. No specific antiviral therapy is currently available. Therefore, only supportive measures are indicated; abstinence from alcohol may be advised. The subjective impression of the patient should guide the doctor’s approach. No measures such as quarantine, long-term hospitalization, vitamin administration (except in the case of diagnosed avitaminosis), other “supportive” medications, or blood transfusions should be administered to patients.
4. Acute hepatitis B
4.1 Pathogenesis and natural course
Hepatitis B virus (HBV) is a DNA-containing virus of the Hepadnaviridae family. The virus is present in most body fluids in individuals with acute or chronic hepatitis and in inactive carriers. It is transmitted parenterally — for example, as a result of sharing needles. Oral transmission is unlikely. Sexual contact is a frequent cause. Unvaccinated health-care workers are a high-risk group due to the risk of needlestick injury. In endemic areas, hepatitis B is often transmitted vertically or horizontally amongst young children playing together (through biting and scratching).
The incubation period for HBV is 60 days, and it can vary from 28 to 160 days.
Approximately 30% of infections among adults present as icteric hepatitis, and 0.1–0.5% of patients develop fulminant hepatitis. When fulminant hepatitis occurs, the immune response to infected hepatocytes is overwhelming and there is often no evidence of viral replication. Testing for HBsAg may be negative; there is therefore a need for further anti-HBc (IgM) testing.
Infection resolves in over 95% of adults with loss of serum HBsAg and the subsequent the appearance of anti-HBs. Natural immunity is characterized by the detection of anti-HBc plus anti-HBs.
HBV infection leads to one of four outcomes:
- Recovery after acute infection (> 95% in previously healthy adults over the age of 40)
- Fulminant hepatitis
- Chronic hepatitis B
- Inactive carrier state
The outcome of HBV infection depends on immunological factors and possibly in part on the characteristics of the virus. The age at which the infection occurs plays a very important role. When it is acquired perinatally and in infants under 1 year of age, chronic infection will develop in 80–90% of cases; in children between the ages of 1 and 5, 30–50% will go on to develop chronic infection. By comparison, 30–50% of adults who become actively infected with HBV are symptomatic, but only 2–6% of these adults develop chronic infection. Some 95–99% of previously healthy adults with an acute HBV infection recover completely.
4.2 Epidemiology: prevalence and incidence
The World Health Organization (WHO) estimates that over 5 million cases of acute hepatitis B infection occur annually. The incidence of HBV infection and patterns of transmission vary greatly throughout the world, depending on local endemicity, with rates between 0.1 and 120 per 100,000.
The average annual incidence of acute hepatitis B in Europe is 20 per 100,000 population. The resulting estimate is approximately 950,000 people infected every year. Of these, 90,000 will become chronically infected, of whom 20,000 will die of cirrhosis or liver cancer.
Hepatitis B virus infection is a global health problem. Two billion people have been infected worldwide; 360 million are chronically infected; over 520,000 die each year (50,000 of acute hepatitis B and 470,000 of cirrhosis or liver cancer).
The prevalence and incidence of HBV varies widely in different areas of the world. It is most prevalent in China, south-east Asia, sub-Saharan Africa, most Pacific islands, and the Amazon basin. There are also high prevalence rates in the circumpolar region (north pole).
In the developed world, hepatitis B is an illness that mainly affects high-risk adults; in the world’s poorer areas, it is highly endemic and widely present in children. All vaccination programs are best focused on immunization in infants and children, because at this age chronic infection can be prevented.
In the Western world, the focus has been on vaccinating adults in high-risk groups such as male homosexuals and health-care workers. The WHO advocates universal vaccination.
4.3 Risk factors and transmission
Adult risk groups for acute HBV infection are:
- Health-care workers
- Policemen
- Migrant populations
- Asylum seekers and refugees
- Military personnel
- Tourists and students (unsafe sex)
- Any unsafe sex
- Injecting drug users
- Hemodialysis patients (always become carriers)
- Firemen
- People in prison
- Persons receiving unsafe injections
- Persons receiving unscreened blood transfusions
- Unsafe piercing, tattooing, acupuncture, tribal scarification, circumcision
Reuse of injection equipment may cause up to 20 million HBV infections each year.
Transmission routes. HBV is transmitted through body fluids such as blood, saliva, and semen (it is controversial whether there is transmission through breast milk). The route may be:
- Perinatal (from mother to baby at birth; vertical)
- From child to child (horizontal)
- From unsafe injections and transfusions (parenteral)
- Nonsterile instruments, tattoo needles, dental equipment, other sharp objects used, for example, in scarification or female circumcision
- Sexual contact
- Unprotected sex (whether heterosexual or homosexual)
HBV is transmitted through either skin puncture or mucosal contact with blood or other infectious body fluids. The virus is found in highest concentrations in blood and serous exudates.
4.4 Diagnosis and differential diagnosis
The most common symptom of hepatitis infection is fatigue or tiredness. Fever, muscle aches, joint aches, and serum sickness syndrome may be present in the prodromal stage of acute HBV. Some people will notice that their urine becomes darker, and their skin will show a yellowish tint (jaundice).
- Principal symptoms:
- Fatigue and tiredness
- Malaise
- Jaundice
- Fever
- Muscle and joint aches
- Less common symptoms:
- Weight loss
- Depression
- Anxiety, irritability
- Headaches
- Sleep disturbance
- Discomfort in the abdomen on the right side
- Itching
- Nausea and diarrhea
- Appetite loss
Serological tests are available commercially for a variety of antigens and antibodies associated with HBV infection. In a suspected acute infection, HBsAg will be positive and should have cleared within 3–6 months after the acute onset (a follow-up re-check should always be carried out). It is impossible to distinguish acute hepatitis B from a flare-up of chronic hepatitis B without follow-up — this is essential in all cases.
4.5 Acute hepatitis B management
Spontaneous recovery occurs after acute infection with HBV occurs in 95–99% of previously healthy adults. Antiviral therapy is not therefore likely to improve the rate of recovery and is not required unless the disease is accompanied by a nonhepatic complication such as periarteritis nodosa. In such cases, and in immunocompromised individuals (e.g., those with chronic renal failure), antiviral therapy with lamivudine may be recommended.
In fulminant hepatitis, meticulous intensive care may improve the survival, but orthotopic liver transplantation is the only therapy that has been shown to improve patient outcomes.
Full recovery with development of anti-HBs provides long-term protection.
4.6 Prevention
Vaccination (available since the early 1980s) continues to be the best way for dealing with the condition. Hepatitis B is preventable, and universal vaccination is probably best — certainly in countries with a high prevalence.
Two types of hepatitis B vaccine are available:
- Recombinant or genetically engineered vaccines are made using HBsAg synthesized in yeast (Saccharomyces cerevisiae) or in mammalian cells into which the HBsAg gene has been inserted. Both consist of a suspension of HB surface antigen. Each country has different preparations.
- Human plasma-derived vaccines (PDVs) are prepared from purified HBsAg from the plasma of individuals with chronic HBV infection. There are more than 15 different PDVs licensed throughout the world.
There are no significant differences between the two types of vaccine with regard to safety, immunogenicity, or efficacy.
HBV vaccines will generate protective levels (> 10 IU/mL) of antibodies to HBsAg in 95% of children and 90% of adults. Revaccination is effective in 80% of those who do not respond to the primary vaccination. Revaccination of nonresponders is not recommended after two series of vaccinations (six doses). A distinction can be drawn between preexposure and postexposure vaccination.
4.6.1 Preexposure vaccination
This is especially relevant in high-risk groups. There are a number of recombinant vaccines with similar efficacy, although the dosage may differ — for example:
Recombivax HB (10 μg of HBsAg)
- Child < 11 y with an HBsAg-negative mother: 2.5 μg (babies at birth)
- Child < 11 y with an HBsAg-positive mother: 5 μg
- Child 11–19: 5 μg
- Immunocompetent adult: 10 μg
- Immunosuppressed person: 40 μg
- Renal dialysis patient: 40 μg
Engerix-B (20 μg of HBsAg)
- Child < 10 y: 10 μg (babies at birth)
- Child > 10 y: 20 μg
- Adult: 20 μg
- Immunosuppressed person: 40 μg
- Dialysis patient: 40 μg
4.6.2 Postexposure vaccination
A combination of hepatitis B immunoglobulin (HBIg), when available, and HBV vaccine is recommended. If HBIg is available (in most countries it is not), it should be given to all children of HBs-positive mothers at the time of delivery. This is particularly important in neonates, in whom an immediate start of postexposure immunization will prevent infection in infants of HBV-infected mothers. It is important to vaccinate within 24 hours. There is no evidence of a protective effect if the vaccine is given more than 7 days after delivery.
Direct exposure (percutaneous inoculation or transmucosal exposure) to HBsAg-positive body fluid (e.g., needlestick injury):
- HBIg single intramuscular dose of 0.06 mL/kg (as soon as possible)
- Followed by a complete course of HBV vaccination (initiated within 7 days)
Direct exposure following sexual contact with an individual with HBV:
- HBIg single intramuscular dose of 0.06 mL/kg (within 14 days; very expensive and not affordable in most places)
- Accompanied by a complete course of HBV vaccination (do not wait!).
4.6.3 Contraindications and side effects
There are very few contraindications:
- Severe allergic reaction to previous doses
- If the patient has a severe allergic reaction to baker’s yeast, plasma-derived HBV vaccine can be used instead
- Fever > 38.5 °C
The following are not contraindications to administering HBV vaccine:
- Any minor illness, such as respiratory tract infection or diarrhea with a temperature below 38.5 °C
- Allergy or asthma
- Treatment with antibiotics
- Human immunodeficiency virus (HIV) infection; however more information is needed on the efficacy of HBV vaccination in neonates or infants who are infected with HIV.
- Breastfeeding
- History of seizures
- Chronic illnesses
- Stable neurological conditions
- Prematurity or low birth weight
- History of jaundice at birth
- Pregnancy (although not generally recommended)
4.6.4 Vaccine safety and quality
Thermostability:
- HBV vaccines should never be frozen. The freezing point of HBV vaccine is –0.5 °C.
- HBV vaccine is stable for at least 4 years if stored at 2–8 °C.
- HBV vaccines are relatively heat-stable and only have a small loss of potency when stored for 2–6 months at a temperature of 37 °C.
The shake test: if you see HBV vaccine frozen, then it is damaged. However, a vaccine may also have been frozen earlier and then thawed again. The shake test can be used to check whether the vaccine has been damaged by earlier freezing. Compare the vaccine that you suspect has been frozen and thawed with vaccine from the same manufacturer that you are sure was never frozen.
- Shake the vaccine vials.
- Look at the contents carefully.
- Leave the vaccines to stand side by side for 15–30 minutes for any sediment to settle.
- Do not use it if a sediment settles below an almost-clear liquid.
4.7 The future
More potent adjuvants may reduce the number of vaccinations from three to two or even one jab. New childhood vaccines are currently being developed.
4.8 Further information
4.9 Summary – sequence of serum testing in a suspected case of acute hepatitis
- Always test for HAV IgM
- In Asia, add a test for HEV (if available — possibly a diagnosis of exclusion)
- If a risk factor is present, test for HBsAg and for HCV RNA (plus anti-HCV)
- Test sequence:
- HBc IgM and HBsAg
- HAV IgM
- Anti-HCV
- HCV RNA
- Anti-HEV (only if the above are negative)
5. Acute hepatitis C
5.1 Pathogenesis and natural course
Hepatitis C virus (HCV) is an RNA-containing virus of the Flaviviridae family. The incubation period varies from 14 to 160 days, with a mean of 7 weeks. Most acute and chronic infections are asymptomatic. If symptoms occur, they usually last 2–12 weeks. The lack of a strong T-lymphocyte response is responsible for the high rate of chronic infection. Anti-HCV is not protective (nonneutralizing antibodies). Unlike the other forms of acute viral hepatitis, acute HCV is very likely to become chronic. This underlines the importance of finding ways of preventing the condition from becoming chronic. Standard monotherapy with interferon alpha reduces the progression to chronic HCV to < 10%. Unfortunately, most acute infections are missed as they are asymptomatic, and the opportunity to treat is therefore rare.
5.2 Epidemiology: prevalence and incidence
The global incidence of hepatitis C is currently unknown. Preliminary estimates of the incidence of HCV suggest that 6,400,000 HCV infections may occur each year. In the United States, it is estimated that more than 20,000 cases occur each year.
Fig. 4 HCV infections per 100,000: preliminary estimates
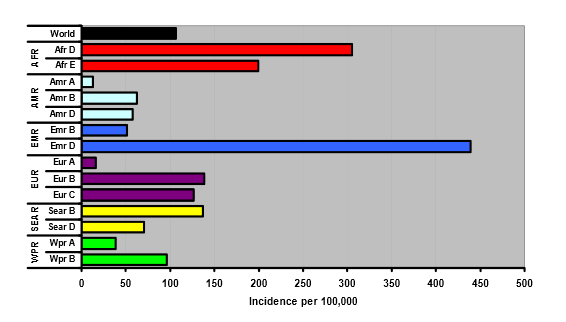
WHO regions: AFR, Africa; AMR, America; EMR, eastern Mediterranean region; EUR, Europe; SEAR, south-east Asia region; WPR, western Pacific region.
The incidence of new symptomatic infections has been estimated to be one to three cases per 100,000 persons annually, but rates of more than 20 per 100,000 have been reported. The actual incidence of new infections is obviously much higher (the majority of cases being asymptomatic). The incidence is declining for two reasons — firstly, transmission via blood products can be reduced to near zero; and secondly, universal precautions have reduced transmission in medical settings.
Fig. 5 HCV epidemiology: attributable proportions (%)
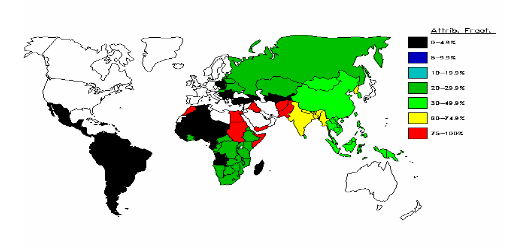
Approximately 3% of the world’s population is infected with HCV — a total of 170 million people. In the U.S., about 4 million people are infected with HCV, of whom it is estimated that 2.7 million have chronic infection.
5.3 Risk factors and transmission
Transmission is from blood to blood. As a blood-borne infection, HCV may potentially be transmitted sexually, mainly in individuals with other sexually transmitted diseases. The perinatal transmission rate is around 5%, much lower than the rates for HIV and HBV. Breastfeeding does not pose a risk. Health-care workers are at risk, mostly due to nosocomial transmission (needlestick injury carries a 3% HCV risk). Also at risk are individuals in prisons and persons born in countries with high rates of endemic disease.
The CDC has listed the risk groups and categories shown in Table 1.
Table 1
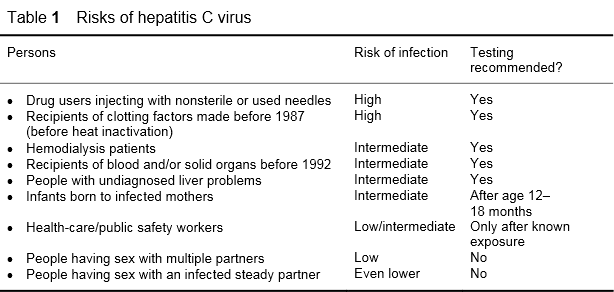
The nosocomial risk is about 1% — associated with endoscopy, multiple-dose vials,and surgery. Individuals with sexually transmitted diseases (STD), including such common ones as herpes, represent an additional risk group. Other potential risk activities include cocaine snorting, tattoos, body piercing, iatrogenic causes (contaminated equipment), tribal scarification, and mass circumcision ceremonies.
5.4 Diagnosis and differential diagnosis
After initial exposure, HCV RNA can be detected in blood within 1–3 weeks. Antibodies to HCV are detected by enzyme immunoassay (EIA) only in 50–70% of cases when symptoms begin, with the rate rising to more than 90% after 3 months.
Liver cell injury becomes evident after 4–12 weeks with elevated ALT levels. Acute infection can be severe, but it is rarely fulminant. Studies show that the risk of fulminant hepatitis is very low, at < 1%.
The most widely used screening tests for HCV are based on enzyme-linked immunosorbent assays (ELISA, EIA). HCV can be diagnosed by the presence of anti-HCV in serum, but antibody tests often do not give positive results for up to 3 months after acute infection. Testing for HCV RNA is the best method of diagnosing acute HCV, particularly if it is then followed by the development of anti-HCV, with seroconversion observed.
Polymerase chain reaction (PCR) tests detect HCV RNA in serum within 1–2 weeks after infection. ELISA for antibody testing has a 97% sensitivity rate.
The antibody may be undetectable for up to 8 weeks after infection, and acute HCV infection is usually subclinical (the “window phase”). The antibody does not confer immunity.
An assay prototype designed to detect and quantify total hepatitis C virus (HCV) core antigen (HCVcoreAg) protein in serum and plasma, in the presence or absence of anti-HCV antibodies, has recently been developed. Investigations show that HCVcoreAg testing makes it possible to detect HCV infection about 1.5 months earlier than the HCV Ab screening tests and an average of only 2 days later than quantitative HCV RNA detection in individual specimens.
5.4.1 Blood tests for acute HCV
Anti-HCV
Note: Anti-HCV does not clarify whether the infection is new (acute), chronic, or no longer present.
Qualitative tests for detecting the presence or absence of virus (HCV RNA):
- Generic polymerase chain reaction
- Amplicor HCV
Quantitative tests for detecting the titer of virus (HCV RNA):
- Amplicor HCM Monitor
- Quantiplex HCV RNA (bDNA)
- TMA (most sensitive)
5.5 Acute HCV management
Indication for treatment: seroconversion to serum HCV RNA–positive or HCVcoreAg-positive. Early identification of HCV is important, because there is evidence that early intervention with standard interferon alpha can markedly reduce the risk of chronic infection from 80% to 10%. There is no preexposure prophylaxis for HCV.
5.6 Prevention
Immunoglobulins are ineffective in preventing HCV. There is no really effective passive or active immunization. Behavioral changes and limiting exposure to high-risk situations offers the best chance of primary prevention.
5.7 The future
Prevention. An important goal is the development of a HCV vaccine that induces cell-mediated immunity. Both therapeutic and prophylactic vaccines are currently in the early stages of development.
Treatment for acute HCV. Future studies should be larger and more evidence-based and they should focus on the efficacy of peginterferons and the time when therapy should be started:
- Start therapy immediately after diagnosis; or
- Delay the start of therapy for 2–4 months to avoid treating those who would recover spontaneously (only in 10–30% of cases).
5.8 Further information
6. Acute hepatitis D
6.1 Pathogenesis and natural course
HDV only co-occurs with HBV. Hepatitis D virus (HDV) is a defective single-stranded RNA virus of the Deltaviridae family. It is an incomplete RNA virus that needs the hepatitis B surface antigen to transmit its genome from cell to cell. It therefore only occurs in people who are positive for the hepatitis B surface antigen. The mean incubation period varies from 60 to 90 days, but it can vary as widely as 30–180 days.
Because HDV relies absolutely on HBV, the duration of the HDV infection is entirely determined by the duration of the HBV infection. HBV replication is suppressed in most HDV-infected individuals. HDV infection can occur either as a co-infection with HBV or as a superinfection in those with chronic HBV.
- Co-infection:
- Severe acute disease
- Low risk of chronic infection
- Indistinguishable from acute HBV
- Superinfection
- Usually develops acute exacerbation of chronic hepatitis
- High risk of chronic liver disease
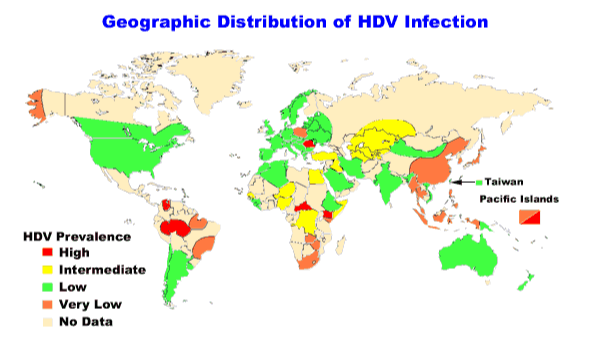
6.2 Epidemiology: prevalence and incidence
There is a decreasing prevalence of both acute and chronic hepatitis D in the Mediterranean area and in many other parts of the world, which has been attributed to a decline in the prevalence of chronic HBsAg carriers in the general population. The incidence of HDV in the general Italian population declined from 3.1 per 100,000 population in 1987 to 1.2 per 100,000 in 1992. However, new foci of high HDV prevalence are continuing to be identified, as in the case of the island of Okinawa in Japan and areas of China, northern India, and Albania. Global incidence figures for the prevalence are not available.
Fig. 6 HDV epidemiology
6.3 Risk factors and transmission
Modes of transmission:
- Percutaneous exposure (injecting drug users)
- Permucosal
- Sexual contact
The mode of transmission of HDV is similar to that of HBV.
The risk of fulminant hepatitis in co-infection is 5%. Otherwise, the prognosis of co-infection is generally good. The prognosis for superinfection is variable. There is some suggestion that the chronic liver disease is more severe, but this is not universally the case.
6.4 Diagnosis and differential diagnosis
During acute HDV infection, HDV Ag and HDV-RNA (PCR) appear early, and anti-HDV of the IgM class appears later. It may take 30–40 days after the first symptoms appear before anti-HDV can be detected.
6.4.1 Serology in the case of co-infection
The serologic course of HDV infection varies depending on whether the virus is acquired as a co-infection with HBV or as a superinfection in a person who is already a hepatitis B carrier. In most individuals with HBV–HDV co-infection, both IgM antibody to HDV (anti-HDV) and anti-HDV IgG are detectable during the course of infection. However, in about 15% of patients, the only evidence of HDV infection may be the detection of either anti-HDV IgM alone during the early acute period of illness or anti-HDV IgG alone during convalescence. Anti-HDV generally declines to subdetectable levels after the infection resolves, and there is no serologic marker that persists to indicate that the patient was ever infected with HDV. Hepatitis delta antigen (HDAg) can be detected in serum in only about 25% of patients with HBV–HDV co-infection. When HDAg is detectable, it generally disappears as HBsAg disappears, and most patients do not develop chronic infection. Tests for anti-HDV IgG are no longer commercially available in the United States or Europe, so it is not possible always to confirm the diagnosis.
Tests for IgM anti-HDV, HDAg, and HDV-RNA by PCR are only available in research laboratories.
6.4.2 Serology in the case of superinfection
In patients with chronic HBV infection who are superinfected with HDV, several characteristic serologic features generally occur, including:
- The titer of HBsAg declines at the time HDAg appears in the serum.
- HDAg and HDV RNA remain detectable in the serum, because chronic HDV infection generally occurs in most patients with HDV superinfection, in contrast to the case with co-infection.
- High titers of both IgM and IgG anti-HDV are detectable, which persist indefinitely. HBV replication is usually suppressed.
6.5 Acute hepatitis D management
No specific treatment is available. Some success has been reported with the viral DNA polymerase inhibitor foscarnet.
6.6 Prevention
- HBV–HDV co-infection is prevented by vaccination against HBV.
- Preventing HBV infection prevents HBV–HDV superinfection.
- Education to reduce risk behaviors in individuals with chronic HBV infection.
HDV co-infection can be prevented with HBV preexposure or postexposure prophylaxis.
6.7 The future
It is unknown whether a new delta antigen test will be developed.
6.8 Further information
7. Acute hepatitis E
7.1 Pathogenesis and natural course
Hepatitis E virus (HEV) is an RNA-containing virus of the Caliciviridae family. The key feature is that, like HAV, it is self-limiting. Management of HEV should therefore be supportive. Hospital admission and medication are not generally necessary, except for pregnant women and those with background chronic liver disease.
The average HEV incubation time is 40 days, and it can vary from 15 to 60 days. The overall mortality rate is 1–3%. In pregnant women it is 15–25%. The clinical presentation of HEV is the same as for HAV. HEV infection is self-limiting and does not require treatment.
7.2 Epidemiology: prevalence and incidence
Fig. 7 HEV epidemiology
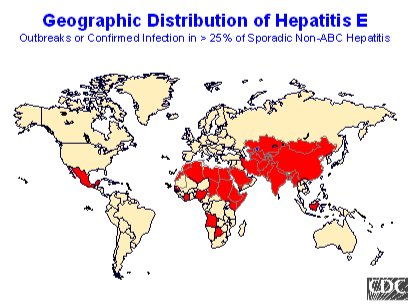
Outbreaks of hepatitis E have occurred over a wide geographic area, primarily in developing countries with inadequate sanitation. The reservoir of HEV in these areas is unknown. The occurrence of sporadic HEV infections in humans may maintain transmission during interepidemic periods, but a nonhuman reservoir for HEV is also possible. In the United States, Europe, and other nonendemic areas, where outbreaks of hepatitis E have not been documented, a low prevalence of anti-HEV (< 2%) has been found in healthy populations. The source of infection for these persons is unknown. (N.B. the map of HEV infection generalizes the available data, and patterns may vary within countries.)
7.3 Risk factors and transmission
HEV is transmitted primarily by the fecal–oral route, and fecally contaminated drinking water is the most frequent cause of transmission. Transmission may occur vertically. Transmission between persons is minimal. Blood-borne transmission has not been demonstrated.
HEV has been implicated in large epidemics in Asia, Africa, and Mexico. Young adults (aged 20–40) are affected most frequently. The prognosis is generally good, except in pregnant women. Preexposure prophylaxis for HEV is not available.
7.4 Diagnosis and differential diagnosis
Assays have been developed for HEV antigen and IgM/IgG antibody testing, but they are not widely available commercially. The virus can be identified in feces, bile, and hepatocyte cytoplasm. Serologic testing for HEV is available.
Further information is available from the Centers for Disease Control and National Institutes of Health in the US (http://www.cdc.gov/).
7.5 Acute hepatitis E management
Treatment is supportive only. Pregnant women are a special risk category. Pregnant women with acute hepatitis E infection have an approximately 15% risk of fulminant liver failure. The mortality rate is high, ranging from 5% to 25% in different studies. HEV infection causes mortality in up to 25% of pregnant women in the third trimester of pregnancy.
7.6 Prevention
The best way of avoiding HEV infection is to avoid using untreated drinking water. Drinking water or eating ice of unknown purity, uncooked shellfish, and foods washed in drinking water should be avoided. Currently, there is no commercially available vaccine for HEV.
7.7 The future
Safe drinking water and a good sanitation infrastructure are the keys to eradicating HEV infection.
7.8 Further information
7.9 Summary
Hepatitis E occurs in annual epidemics, often during the rainy season, and is mainly associated with fecally contaminated drinking water; exceptions are food-borne epidemics (raw or uncooked shellfish). Epidemic and sporadic cases have been reported from south-east and central Asia, the Middle East, northern and western Africa, and North America. Epidemics of hepatitis E are more common in parts of the world with hot climates and are rare in temperate climates. Preliminary evidence indicates that up to 40% of cases of acute hepatitis in Egypt and India are due to HEV and that hepatitis E is responsible for up to 70% of acute hepatitis cases in countries such as Saudi Arabia, Vietnam, Indonesia, Malaysia, and Nepal.
8. References
EASL Jury. EASL International Consensus Conference on Hepatitis B, 13–14 September, 2002: Geneva, Switzerland. Consensus statement (short version). J Hepatol 2003;38:533–40 (http://www.easl.ch/hbv2002/01630170.pdf).
Lavanchy D. Public health measures in the control of viral hepatitis: a World Health Organization perspective for the next millennium. J Gastroenterol Hepatol 2002;17(Suppl):S452–9.
National Institutes of Health consensus paper: http://consensus.nih.gov/2002/2002HepatitisC2002116html.htm.
Ryder SD, Beckingham IJ. ABC of diseases of liver, pancreas, and biliary system: acute hepatitis. BMJ 2001;322:151–3 (PMID: 11159575)
9. Useful web sites
10. Queries and feedback
The Practice Guidelines Committee welcomes any comments and queries that readers may have. Do you feel we have neglected some aspects of the topic? Do you think that some procedures are associated with extra risk? Tell us about your own experience. You are welcome to click on the link below and let us know your views.
guidelines@worldgastroenterology.org











