Endoscopic Surgery – An Evolving Interdisciplinary Subspecialty!
Vol. 27, Issue 2 (June 2022)
 |
Amol Bapaye, MD, MS, FASGE, FJGES, FISG, FSGEI
Director, Shivanand Desai Center for Digestive Disorders
Deenanath Mangeshkar Hospital and Research Center
Erandwane, Pune, India
|
Although gastrointestinal (GI) endoscopy made humble beginnings in 1958 as a diagnostic tool, intraluminal therapeutic interventions through the endoscope have evolved significantly. Since the first description of polypectomy by Shinya et al. in 1969,1 endoluminal interventions made tremendous progress in the subsequent years by the development of techniques like endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD).2, 3 Despite these developments, for a long time, endoscopic interventions were limited to treatment of intraluminal pathologies, or to the so called “first space.”
The tremendous development and technological explosion of laparoscopic surgery in the late 1980s and 1990s stimulated pioneers in flexible endoscopy to aim to perform “no scar” flexible endoscopic surgery within the peritoneal space. This resulted in the development of natural orifice transluminal endoscopic surgery (NOTES) as a specialty in 2004, wherein transgastric or transvaginal approaches were used to gain access to the peritoneal cavity.4, 5 Although NOTES remained largely a proof-of-principle paradigm, the concept provided endoscopists with the opportunity to explore the possibilities of accessing the peritoneal or “second space” reliably and safely. Secure closure of the bowel wall defect was one of the most important challenges for the development and application of NOTES. Easier or safer modes to access the peritoneal cavity were therefore explored.
Sumiyama et al. in 2007 described the concept of submucosal tunneling endoscopy using a mucosal flap valve (SEMF) to access the peritoneal cavity.6 The submucosal, or “third space” as it is called, provides a unique opportunity to access the deeper (intramural) layers of the GI tract as well as acts as a port to access the peritoneal cavity. Pasricha et al. evaluated the concept of submucosal myotomy in porcine models.7 Inoue and colleagues performed the first peroral endoscopic myotomy (POEM) for achalasia cardia in 2008. They published their “first in the world” case series of 17 patients, unleashing a decade of research, evolution, and advancement in the field of third space endoscopy.8 In this article, we discuss the current applications for this exciting specialty and future directions.
Endoscopic Submucosal Dissection (ESD) – The Precursor
ESD is the first procedure that visualized and required dissection in the submucosal space. ESD was conceived to address the limitations posed by EMR to perform enbloc resection for mucosal neoplasia larger than 20 mm in size. ESD has been extensively used for the treatment of early gastric cancer and mucosal lesions, esophageal squamous cell carcinoma, and early colorectal cancer and dysplastic lesions.9 Results of ESD have consistently demonstrated high success rates of enbloc and R0 resection r and relatively low recurrence rates and adverse events.
Third Space or Submucosal Endoscopy
Submucosal endoscopy using a mucosal flap valve (SEMF), also called third space endoscopy (TSE), submucosal surgery, or submucosal endoscopic surgery, is a unique concept (Figure 1). It utilizes the mucosal flap valve principle to create a tunnel in the submucosal layer through a proximally placed mucosal incision. This access route is then used to operate in the deeper layers of the gastrointestinal (GI) tract or to gain access to the peritoneal or mediastinal cavity, after which the incision is closed using endoscopic clips or sutures. The advantage of such an approach is the added procedural safety because of the stepwise dissection of layers of the bowel wall that separates the mucosal incision and myotomy sites.10, 11
Figure 1 Third Space Endoscopy Procedure Steps
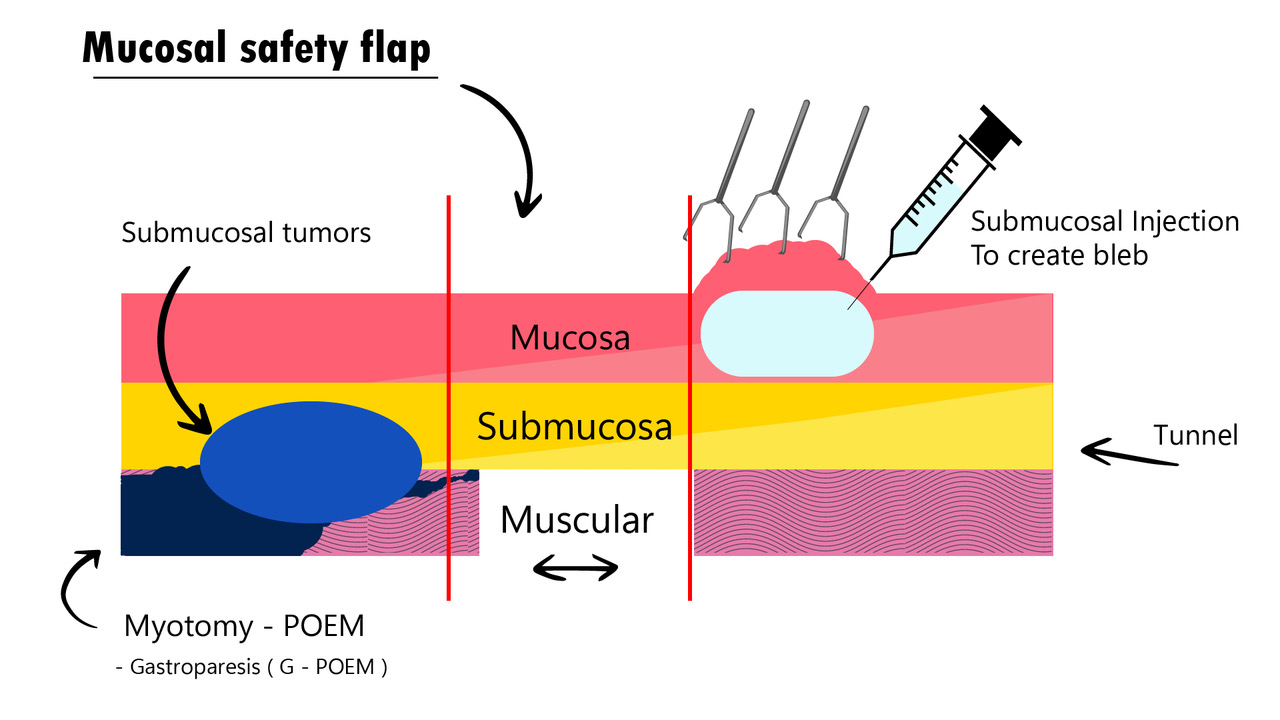
Peroral endoscopic myotomy (POEM) is the index procedure first performed using TSE principles. After the unprecedented success and popularity of POEM, endoscopists around the world have extended the applications of TSE to other spastic disorders of the GI tract. In addition, subepithelial tumors of the GI tract are being resected using submucosal tunneling endoscopic resection (STER) or endoscopic full-thickness resection (EFTR) procedures.12, 13
The general technique for TSE procedures involves creating a submucosal cushion proximal to the area of interest in the bowel, mucosal incision over the cushion, entry into the submucosa, creating a longitudinal tunnel along the axial length of the organ of interest, accessing the muscle layer distal to the site of mucosal entry, and performing the desired intervention—myotomy, tumor resection, etc., followed by endoscopic closure of the mucosal incision using clips or sutures. General anesthesia and carbon dioxide insufflation are mandatory while performing these procedures. Apart from standard high-definition endoscopes, a variety of instruments such as endoscopic caps, knives, coagulation graspers, clips, and suturing devices are used while performing these procedures.
TSE procedures are largely safe, and most procedures have reported a low incidence of adverse events. Pneumoperitoneum (or pneumomediastinum) type adverse events are the most common and occur due to carbon dioxide insufflation while performing the procedure. Using low flow insufflation devices and abdominal or pleural needle decompression have been described as measures to avoid or treat these events, respectively. Intraprocedural (or delayed) bleeding, perforation, and infection have all been reported, and care must be taken to adhere to surgical principles of hemostasis, closure, and disinfection to avoid or minimize the occurrence of such events. Severe adverse events are rare, although mortality has been anecdotally reported.10, 11
POEM for Achalasia Cardia
POEM is the oldest and most evaluated of all TSE procedures (Figure 2). It has the distinction of being the procedure that gained fastest popularity and acceptance in the history of endoscopy. Based on the current available literature on POEM, an estimated more than 25,000 POEM procedures have been performed worldwide. POEM has demonstrated excellent technical and clinical success rates for treatment of all types of achalasia cardia and has reported consistently low adverse events. Success rates above 90% have been consistently reported in most studies and meta-analyses. POEM has demonstrated excellent outcomes for naïve achalasia as well as for prior treatment failures. It has shown excellent outcomes in the pediatric population as well. While comparing POEM with other modalities, it has demonstrated superior results over endoscopic pneumatic balloon dilatation (EBD), whereas outcomes for laparoscopic Heller’s myotomy (LHM) and POEM are comparable for achalasia subtypes I and II. POEM has been successfully implemented for the treatment of type III achalasia and other spastic esophageal disorders (SEDs) like diffuse esophageal spasm (DES), GE junction outflow obstruction, and jackhammer esophagus (JH). Because POEM is performed through the esophageal lumen, it has the advantage that a customized length of myotomy can be performed, depending on the underlying pathology. Results of POEM in type III achalasia and SEDs have demonstrated significantly higher success rates as compared to LHM or EBD.10, 11
Figure 2 POEM Procedure Steps
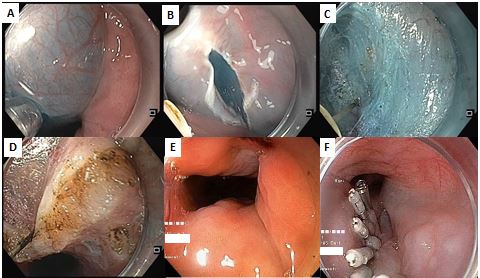
Patients with achalasia cardia being subjected to POEM require prior evaluation by EGD and high-resolution manometry to confirm and classify the type of achalasia. POEM is usually performed as an in-patient procedure or more recently has also been safely performed on an out-patient basis.
Submucosal Tunneling Endoscopic Resection (STER) for GI Subepithelial Tumors
Subepithelial lesions (SELs) of the GI tract can arise from the submucosal or muscularis propria layer of the bowel and include a variety of pathologies – leiomyoma, gastrointestinal stromal tumors (GISTs), lipoma, neuroendocrine tumors (NETs), neural origin tumors like schwannoma, etc.
A tissue diagnosis of these lesions is often challenging and, although most of these lesions are benign, some such as GISTs and NETs harbor malignant potential. Resection is therefore often mandated, especially when the lesion is known to be premalignant, has demonstrated rapid interval growth, and/or is symptomatic. Using the TSE principles, these tumors, particularly those in the esophagus, gastroesophageal junction (GEJ), or stomach, can be resected endoscopically (Figure 3). As in POEM, a submucosal tunnel is created through a proximally placed mucosal incision, and the tumor is resected (enucleated) from the muscle layer. Since the defects in the muscle and mucosa are at different locations, full-thickness perforation is avoided and safety is enhanced.12
Figure 3 STER Procedure Steps
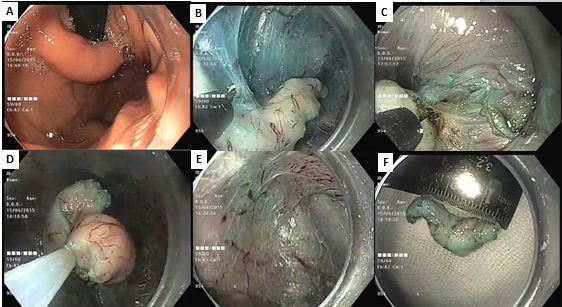
Outcomes of STER demonstrated high clinical efficacy with excellent complete resection rates and high enbloc resection rates in two large meta-analyses. Adverse events were reported more frequently as compared to POEM (14.8%), but most were minor in nature. Risk factors for inferior outcomes and adverse events following STER have been defined as SELs with diameter > 3.5 cm, overlying ulcerated mucosa, SELs with irregular borders, lesions in the fundus or on the gastric lesser curvature, tumors arising from the deep portion of the muscularis propria layer, long procedure time, and insufflation with air. Compared to surgery (thoracoscopic enucleation), STER has demonstrated significantly superior outcomes for procedure times, length of hospital stay, and cost.10, 11
During STER, the tumor is enucleated from the muscle layer. Although this may be adequate for benign SELs, R0 resection margins can pose a challenge for GIST tumors which have malignant potential. Secondly, STER is technically difficult or nearly impossible to perform in certain areas of the GI tract, like the gastric fundus or lesser curvature. To ensure a wide (oncologically sound) resection margin and to address lesions at these locations, EFTR can be utilized.11
Endoscopic Full-Thickness Resection (EFTR)
EFTR can be performed using one of the three approaches – non-tunneled exposed EFTR, tunneled-exposed EFTR, and non-exposed EFTR – wherein the term “exposed” refers to any temporary exposure of the GI tract lumen to the peritoneal cavity.13 Exposed EFTR involves creation and subsequent closure of bowel wall defects whereas in non-exposed EFTR, the defect is prophylactically closed prior to tumor resection. Tunneled exposed EFTR is like STER, wherein the tumor is resected via a submucosal tunnel and the mucosal incision is closed thereafter. In non-tunneled exposed EFTR, the procedure begins as a standard ESD wherein the tumor is dissected all around from the submucosa, before resecting it from the deeper muscle layers and serosa. The resultant defect must be closed, and this is achieved by various means – standard mucosal clips, combination of endoloop and clips, full-thickness clips or endoscopic suturing. Prototype endoscopic staplers have also been described. The major challenge after non-tunneled exposed EFTR is to achieve safe closure of the defect after resection, which is often challenging due to compromised luminal insufflation after complete tumor resection. With the increasing implementation of endoscopic suturing and retraction methods, these challenges can be somewhat circumvented.
Non-exposed EFTR involves approximation of the serosal surfaces underneath the lesion prior to resection, thereby preventing a full-thickness perforation. A major advance in non-exposed EFTR has been the development of the full-thickness resection device (FTRD), which combines resection with closure using a full-thickness over-the-scope clip (OTSC) in one device. The SEL is grasped and pulled into the transparent distal hood attached to the endoscope, the clip is applied to the base, and the lesion is resected above the clip using an inbuilt snare. Initially described for lower GI conditions, a device for foregut applications has also recently become available. EFTR using FTRD is limited by the size of the lesion and sometimes by the large size of the device itself, which makes its insertion up to the target site technically difficult, particularly through the sigmoid colon or across the cricopharynx. For larger lesions therefore, combined laparoscopic-endoscopic surgery (LECS) has been described.
Majority of EFTRs have been described in the stomach, and for GIST tumors. Although the technique can be applied to other locations in the GI tract, technical difficulties for secure closure limits this applicability. In general, outcomes of EFTR have demonstrated high enbloc and R0 resection rates, low recurrence and adverse events.11
Tunneling Myotomy for Other Spastic GI Conditions
The high success rates of POEM for achalasia prompted endoscopists to devise and perform tunneling endoscopy procedures for other spastic conditions in the GI tract. These include gastric peroral endoscopic myotomy (G-POEM) for refractory gastroparesis,14 tunneling septotomy for Zenker’s or epiphrenic diverticula (D-POEM and Z-POEM respectively)15, 16 and per rectal endoscopic myotomy (PREM) for Hirschsprung disease and acquired megacolon.17
Z-POEM and D-POEM for Esophageal Diverticula
Zenker’s diverticulum is an anatomical outpouching occurring through a defect in the cricopharyngeus muscle at the Killian’s triangle. Conventional treatment has included surgical resection of the diverticulum along with cricopharyngeal myotomy (a procedure with significant morbidity) or endoscopic septotomy. Septotomy, although less invasive, has the inherent risk of recurrence if the septotomy is short and the risk of perforation if it is too long. TSE has enabled safe septotomy to be performed through a submucosal tunnel at the cricopharynx – Z-POEM, wherein an adequate length septotomy can be performed without the risk of perforation and mediastinitis.
Similarly, mid esophageal and epiphrenic diverticula occur in patients often with esophageal motility disorders, particularly achalasia. The same principle is applied to perform diverticular septotomy (D-POEM) for these diverticula. Outcomes of Z-POEM and D-POEM have demonstrated excellent symptom relief and minimal adverse events.
G-POEM for Refractory Gastroparesis
Gastroparesis is caused either because of reduced gastric body contractility or incoordinated peristalsis and opening of the antrum and pyloric sphincter. Typical etiologies are iatrogenic (due to surgical vagal injury), diabetes, or idiopathic. For gastroparesis caused due to pyloric sphincter spasm or incoordinated activity, Khashab et al. performed the first G-POEM or gastric per-oral endoscopic pyloromyotomy.14 The procedure has demonstrated good clinical success rates of approximately 82% in various case series and in meta-analyses. One of the key elements for patient selection for G-POEM is to consider it for those with pylorospasm and avoid in those where gastric hypomotility is the predominant mechanism. In a recent prospective study that critically evaluated follow up parameters, outcomes after G-POEM were reported at a modest 56%.10
PREM for Hirschsprung’s Disease and Megacolon
Hirschsprung’s disease is characterized by aganglionosis of Auerbach and Meissner plexuses starting from the rectum and extending proximally, resulting in a functional obstruction. Using the principles of TSE, per rectal endoscopic myotomy (PREM) has been conceptualized and performed for patients with adult, pediatric and infantile Hirschsprung’s disease (Figure 4). Results of a case series report excellent functional outcomes in terms of relief of constipation, laxative usage, and overall functional bowel scores at medium and long term follow up.18 With more data regarding its outcomes, this procedure promises to be a blessing for patients with Hirschsprung’s disease, wherein until recently surgical pull through was the only alternative and carried significant morbidity.
Figure 4 PREM Procedure Steps
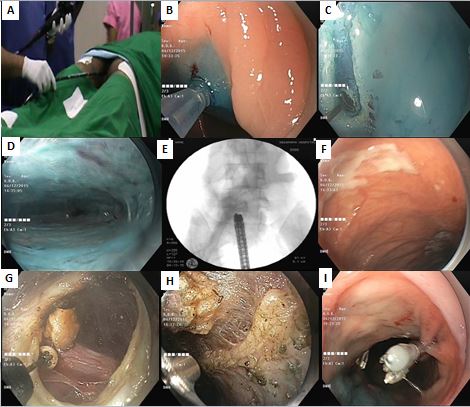
Other Novel Applications and Future Directions
Wagh and Draganov described peroral endoscopic tunneling for restoration of the esophagus (POETRE) for patients with complete esophageal obstruction.19 This technique is a very attractive minimally invasive option for such patients where the alternative is surgery with high morbidity. Submucosal endoscopy has been experimentally used to implant devices within the submucosal space and deep muscle layers. Electrostimulation of the lower esophageal sphincter for treatment of gastroesophageal reflux (GER) has been attempted in animal models.
TSE provides a relatively safe access route to enter the peritoneal or mediastinal cavity. Inoue et al. described the first NOTES anterior partial fundoplication (POEM+F or POEF) performed prophylactically to prevent or to treat post POEM reflux (GER).20, 21 One year follow up results of POEM+F have demonstrated an intact wrap in 83% patients and an impressive control of post POEM GER.22 TSE guided peritoneoscopy or mediastinoscopy have been evaluated in animal studies and have the potential to be used as an alternative to laparoscopy for staging of GI malignancies.
Training in TSE procedures, Future Research, and Perspectives
Most TSE procedures are relatively new. POEM is the most widely studied and its position is reasonably well established within treatment algorithms. For most other procedures, available data is predominantly in form of single or multicenter case series and non-randomized retrospective studies. Few meta-analyses have been published in the last few years; however, because these too are based on single arm case series, the quality of the data is average. The relative rarity of several of these procedure indications pose a further challenge to conduct well designed prospective randomized trials. Hopefully, as TSE enters the second decade of its inception, we can gather more information on these procedures in a prospective fashion.
Training for TSE procedures differs as compared to conventional interventional endoscopy because these procedures are predominantly surgical in nature. There is often a debate as to who should perform these procedures – physician gastroenterologists or surgeons? The answer is not easy. Being surgical procedures, it is logical that surgeons may be more inclined (or better suited) to perform these procedures. However, the majority of global surgical curricula currently provide minimal exposure to flexible endoscopy. Physician gastroenterologists on the other hand, although well trained for flexible endoscopy, lack the surgical mindset (and training) that is often required to undertake these procedures. It is possible that with the advent of these and similar procedures, conventional surgical and endoscopy teaching curricula may undergo appropriate restructuring to provide the (interested) trainees with adequate and optimal instruction and exposure to this specialty.
Credentialing and accreditation for these procedures remains a further challenge. The American and European Societies for Gastrointestinal Endoscopy (ASGE and ESGE) have formulated guidelines for training in ESD and POEM. Although good as a starting point, most of these guidelines require further revision and refinement before they can become applicable at large.
Conclusion
Gastrointestinal endosurgery (GIES) has always been the final frontier for flexible endoscopic interventions. With the advent and rapid development of TSE, this dream has the potential to be fully realized. However, as is true for any new specialty, several challenges have been identified. As TSE enters its adolescence, one can expect that the technique will further evolve, will create excitement within the GI-surgery community, but may also raise several controversies that will require resolution before it matures into a responsible, widely adopted and established adult specialty. Collaboration and partnership between the gastroenterologists and surgical colleagues will be key in advancing this field and ensuring the best outcomes for our patients.
Table 1. Types of Third Space Endoscopic procedures and their clinical applications
|
Disease / Disorder
|
Name of the Procedure
|
Acronym
|
|
Achalasia cardia
|
Peroral endoscopic myotomy
|
POEM
|
|
Refractory gastroparesis
|
Gastric peroral endoscopic myotomy
|
G-POEM
|
|
Zenker’s diverticulum
|
Zenker’s peroral endoscopic myotomy
|
Z-POEM
|
|
Epiphrenic diverticulum
|
Diverticular peroral endoscopic myotomy
|
D-POEM
|
|
Hirschsprung’s disease/acquired megacolon
|
Per rectal endoscopic myotomy
|
PREM
|
|
Subepithelial lesions
|
Submucosal tunneling endoscopic resection/peroral endoscopic tumor resection
|
STER/POET
|
|
Subepithelial lesions
|
Endoscopic full-thickness resection
|
EFTR
|
|
Total esophageal obstruction
|
Peroral endoscopic tunneling for restoration of esophagus
|
POETRE
|
References
1. Wolff WI, Shinya H. Polypectomy via the fiberoptic colonoscope. Removal of neoplasms beyond reach of the sigmoidoscope. N Engl J Med 1973;288:329-32.
2. Tada M, Shimada H, Yanai H, et al. New technique of gastric biopsy. Stomach Intest 1984;19:1107-16.
3. Inoue H, Fukami N, Yoshida T, et al. Endoscopic mucosal resection for esophageal and gastric cancers. J Gastroenterol Hepatol 2002;17:382-8.
4. Kalloo AN, Singh VK, Jagannath SB, et al. Flexible transgastric peritoneoscopy: a novel approach to diagnostic and therapeutic interventions in the peritoneal cavity. Gastrointest Endosc 2004;60:114-7.
5. Khashab MA, Kalloo AN. NOTES: current status and new horizons. Gastroenterology 2012;142:704-710 e1.
6. Sumiyama K, Gostout CJ, Rajan E, et al. Submucosal endoscopy with mucosal flap safety valve. Gastrointest Endosc 2007;65:688-94.
7. Pasricha PJ, Hawari R, Ahmed I, et al. Submucosal endoscopic esophageal myotomy: a novel experimental approach for the treatment of achalasia. Endoscopy 2007;39:761-4.
8. Inoue H, Minami H, Kobayashi Y, et al. Peroral endoscopic myotomy (POEM) for esophageal achalasia. Endoscopy 2010;42:265-71.
9. Pimentel-Nunes P, Dinis-Ribeiro M, Ponchon T, et al. Endoscopic submucosal dissection: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2015;47:829-54.
10. Bapaye A, Korrapati SK, Dharamsi S, et al. Third Space Endoscopy: Lessons Learnt From a Decade of Submucosal Endoscopy. J Clin Gastroenterol 2020;54:114-129.
11. Liu AQ, Chiu PWY. Third space endoscopy: Current evidence and future development. International Journal of Gastrointestinal Intervention 2020;9:42-52.
12. Xu MD, Cai MY, Zhou PH, et al. Submucosal tunneling endoscopic resection: a new technique for treating upper GI submucosal tumors originating from the muscularis propria layer (with videos). Gastrointest Endosc 2012;75:195-9.
13. Committee AT, Aslanian HR, Sethi A, et al. ASGE guideline for endoscopic full-thickness resection and submucosal tunnel endoscopic resection. VideoGIE 2019;4:343-350.
14. Khashab MA, Stein E, Clarke JO, et al. Gastric peroral endoscopic myotomy for refractory gastroparesis: first human endoscopic pyloromyotomy (with video). Gastrointest Endosc 2013;78:764-8.
15. Li QL, Chen WF, Zhang XC, et al. Submucosal Tunneling Endoscopic Septum Division: A Novel Technique for Treating Zenker's Diverticulum. Gastroenterology 2016;151:1071-1074.
16. Maydeo A, Patil GK, Dalal A. Operative technical tricks and 12-month outcomes of diverticular peroral endoscopic myotomy (D-POEM) in patients with symptomatic esophageal diverticula. Endoscopy 2019;51:1136-1140.
17. Bapaye A, Wagholikar G, Jog S, et al. Per rectal endoscopic myotomy for the treatment of adult Hirschsprung's disease: First human case (with video). Dig Endosc 2016;28:680-4.
18. Bapaye A, Dashatwar P, Biradar V, et al. A novel third space endoscopic procedure, per-rectal endoscopic myotomy, for Hirschsprung's disease: Medium and long-term outcomes. Endoscopy 2020.
19. Wagh MS, Draganov PV. Per-oral endoscopic tunneling for restoration of the esophagus: a novel endoscopic submucosal dissection technique for therapy of complete esophageal obstruction. Gastrointest Endosc 2017;85:722-727.
20. Inoue H, Ueno A, Shimamura Y, et al. Peroral endoscopic myotomy and fundoplication: a novel NOTES procedure. Endoscopy 2019;51:161-164.
21. Toshimori A, Inoue H, Shimamura Y, et al. Peroral endoscopic fundoplication: a brand-new intervention for GERD. VideoGIE 2020;5:244-246.
22. Bapaye A, Dashatwar P, Dharamsi S, et al. Single-session endoscopic fundoplication after peroral endoscopic myotomy (POEM+F) for prevention of post gastroesophageal reflux - 1-year follow-up study. Endoscopy 2020.






