Prof. M. Farthing (Chair, United Kingdom)
Prof. M. Salam (Special Advisor, Bangladesh)
Prof. G. Lindberg (Sweden)
Prof. P. Dite (Czech Republic)
Prof. I. Khalif (Russia)
Prof. E. Salazar-Lindo (Peru)
Prof. B.S. Ramakrishna (India)
Prof. K. Goh (Malaysia)
Prof. A. Thomson (Canada)
Prof. A.G. Khan (Pakistan)
Drs. J. Krabshuis (France)
Dr. A. LeMair (Netherlands)
1. Introduction and Epidemiologic Features
According to the World Health Organization (WHO) and UNICEF, there are about two billion cases of diarrheal disease worldwide every year, and 1.9 million children younger than 5 years of age perish from diarrhea each year, mostly in developing countries. This amounts to 18% of all the deaths of children under the age of five and means that more than 5000 children are dying every day as a result of diarrheal diseases. Of all child deaths from diarrhea, 78% occur in the African and South-East Asian regions.
Each child under 5 years of age experiences an average of three annual episodes of acute diarrhea. Globally in this age group, acute diarrhea is the second leading cause of death (after pneumonia), and both the incidence and the risk of mortality from diarrheal diseases are greatest among children in this age group, particularly during infancy – thereafter, rates decline incrementally. Other direct consequences of diarrhea in children include growth faltering, malnutrition, and impaired cognitive development in resource-limited countries.
During the past three decades, factors such as the widespread availability and use of oral rehydration salts (ORS), improved rates of breastfeeding, improved nutrition, better sanitation and hygiene, and increased coverage of measles immunization are believed to have contributed to a decline in the mortality rate in developing countries. In some countries, such as Bangladesh, a reduction in the case fatality rate (CFR) has occurred without appreciable changes in the water supply, sanitation, or personal hygiene, and this can be attributed largely to improved case management.
ORS and nutritional improvements probably have a greater impact on mortality rates than the incidence of diarrhea. Prevailing poor living conditions and insignificant improvements in water, sanitation, and personal hygiene, despite some improvement in nutrition, is perhaps important in explaining the lack of impact on the incidence. Interventions such as exclusive breastfeeding (which prevents diarrhea), continuation of breastfeeding until 24 months of age, and improved complementary feeding (by way of improved nutrition), along with improved sanitation, are expected to affect mortality and morbidity simultaneously. The recommended routine use of zinc in the management of childhood diarrhea, not currently practiced in many countries, is expected to reduce disease incidence.
In industrialized countries, relatively few patients die from diarrhea, but it continues to be an important cause of morbidity that is associated with substantial health-care costs. However, the morbidity from diarrheal diseases has remained relatively constant during the past two decades.
In this guideline, specific pediatric details are provided in each section as appropriate.
2. Causative Agents and Pathogenic Mechanisms
Bacterial agents
In developing countries, enteric bacteria and parasites are more prevalent than viruses and typically peak during the summer months.
Diarrheagenic Escherichia coli. The distribution varies in different countries, but enterohemorrhagic E. coli (EHEC, including E. coli O157:H7) causes disease more commonly in the developed countries.
- Enterotoxigenic E. coli (ETEC) causes traveler’s diarrhea.
- Enteropathogenic E. coli (EPEC) rarely causes disease in adults.
- Enteroinvasive E. coli (EIEC)* causes bloody mucoid (dysentery) diarrhea; fever is common.
- Enterohemorrhagic E. coli (EHEC)* causes bloody diarrhea, severe hemorrhagic colitis, and the hemolytic uremic syndrome in 6–8% of cases; cattle are the predominant reservoir of infection.
Pediatric details. Nearly all types cause disease in children in the developing world:
- Enteroaggregative E. coli (EAggEC) causes watery diarrhea in young children and persistent diarrhea in children with human immunodeficiency virus (HIV).
- Enterotoxigenic E. coli (ETEC) causes diarrhea in infants and children in developing countries.
- Enteropathogenic E. coli (EPEC) causes disease more commonly in children < 2 years, and persistent diarrhea in children.
* EIEC and EHEC are not found (or have a very low prevalence) in some developing countries.
Campylobacter:
- Asymptomatic infection is very common in developing countries and is associated with the presence of cattle close to dwellings.
- Infection is associated with watery diarrhea; sometimes dysentery.
- Guillain–Barré syndrome develops in about one in 1000 of people with Campylobacter colitis; it is thought to trigger about 20–40% of all cases of Guillain–Barré syndrome. Most people recover, but muscle weakness does not always completely resolve.
- Poultry is an important source of Campylobacter infections in developed countries, and increasingly in developing countries, where poultry is proliferating rapidly.
- The presence of an animal in the cooking area is a risk factor in developing countries.
Pediatric details. Campylobacter is one of the most frequently isolated bacteria from the feces of infants and children in developing countries, with peak isolation rates in children 2 years of age and younger.
Shigella species:
- Hypoglycemia, associated with very high case fatality rates (CFRs) (43% in one study) occurs more frequently than in other types of diarrheal diseases
- S. sonnei is common in developed countries, causes mild illness, and may cause institutional outbreaks.
- S. flexneri is endemic in many developing countries and causes dysenteric symptoms and persistent illness; uncommon in developed countries.
- S. dysenteriae type 1 (Sd1) — the only serotype that produces Shiga toxin, as does EHEC. It also is the epidemic serotype that has been associated with many outbreaks during which CFRs can be as high as 10% in Asia, Africa, and Central America. For unexplained reasons, this serotype has not been isolated since the year 2000 in Bangladesh and India.
Pediatric details. An estimated 160 million episodes occur in developing countries, primarily in children. It is more common in toddlers and older children than in infants.
Vibrio cholerae:
- Many species of Vibrio cause diarrhea in developing countries.
- All serotypes (>2000) are pathogenic for humans.
- V. cholerae serogroups O1 and O139 are the only two serotypes that cause severe cholera, and large outbreaks and epidemics.
- In the absence of prompt and adequate rehydration, severe dehydration leading to hypovolemic shock and death can occur within 12–18 h after the onset of the first symptom.
- Stools are watery, colorless, and flecked with mucus; often referred to as “rice-watery” stools.
- Vomiting is common; fever is typically absent.
- There is a potential for epidemic spread; any infection should be reported promptly to the public health authorities.
Pediatric details. In children, hypoglycemia can lead to convulsions and death.
Salmonella:
- Enteric fever — Salmonella enterica serovar Typhi and Paratyphi A, B, or C (typhoid fever); fever lasts for 3 weeks or longer; patients may have normal bowel habits, constipation or diarrhea.
- Animals are the major reservoir for salmonellae. Humans are the only carriers of typhoidal Salmonella.
- In nontyphoidal salmonellosis (Salmonella gastroenteritis), there is an acute onset of nausea, vomiting, and diarrhea that may be watery or dysenteric in a small fraction of cases.
- The elderly and people with immune-compromised status for any reason (e.g., hepatic and lymphoproliferative disorders, hemolytic anemia), appear to be at the greatest risk.
Pediatric details:
- Infants and children with immune-compromised status for any reason (e.g., severe malnourishment) appear to be at the greatest risk.
- Fever develops in 70% of affected children.
- Bacteremia occurs in 1–5%, mostly in infants.
Viral agents
In both industrialized and developing countries, viruses are the predominant cause of acute diarrhea, particularly in the winter season.
Rotavirus:
- Accounts for one-third of diarrhea hospitalizations and 500,000 deaths worldwide each year.
- Associated with gastroenteritis of above-average severity.
Pediatric details:
- Leading cause of severe, dehydrating gastroenteritis among children.
- Nearly all children in both industrialized and developing countries get infected by the time they are 3–5 years of age.
- Neonatal infections are common, but often asymptomatic.
- The incidence of clinical illness peaks in children between 4 and 23 months of age.
Human caliciviruses (HuCVs):
- Belong to the family Caliciviridae—the noroviruses and sapoviruses (previously called “Norwalk-like viruses” and “Sapporo-like viruses.”
- Noroviruses are the most common cause of outbreaks of gastroenteritis, affecting all age groups.
Pediatric details. Sapoviruses primarily affect children. This may be the second most common viral agent after rotavirus, accounting for 4–19% of episodes of severe gastroenteritis in young children.
Adenovirus infections most commonly cause illnesses of the respiratory system.
Pediatric details: depending on the infecting serotype, this virus may cause gastroenteritis especially in children.
Parasitic agents
Cryptosporidium parvum, Giardia intestinalis, Entamoeba histolytica, and Cyclospora cayetanensis: these are uncommon in the developed world and are usually restricted to travelers.
Pediatric details. Most commonly cause acute diarrheal illness in children.
- These agents account for a relatively small proportion of cases of infectious diarrheal illnesses among children in developing countries.
- G. intestinalis has a low prevalence (approximately 2–5%) among children in developed countries, but as high as 20–30% in developing regions.
- Cryptosporidium and Cyclospora are common among children in developing countries; frequently asymptomatic.
Table 1 Overview of causative agents in diarrhea
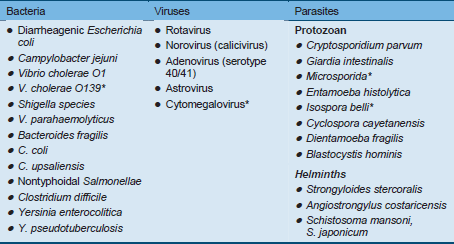
* These agents are no longer reported in the Indian subcontinent.
3. Clinical Manifestations and Diagnosis
Although there may be clinical clues, a definitive etiological diagnosis is not possible clinically (Tables 2–4).
Table 2 Episodes of diarrhea can be classified into three categories

Table 3 Linking the main symptoms to the causes of acute diarrhea—enterohemorrhagic E. coli (EHEC)
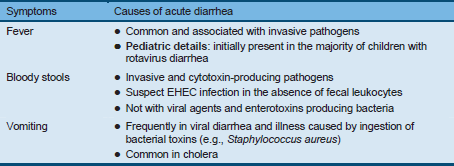
Table 4 Clinical features of infection with selected diarrheal pathogens

Key: ++, common: +, occurs, +/–, variable; –, not common: 0, atypical/often not present.
Clinical evaluation
The initial clinical evaluation of the patient (Table 5) should focus on:
- Assessing the severity of the illness and the magnitude (degree) of dehydration (Table 6)
- Determining likely causes on the basis of the history and clinical findings, including stool characteristics
Table 5 Medical assessment in diarrhea
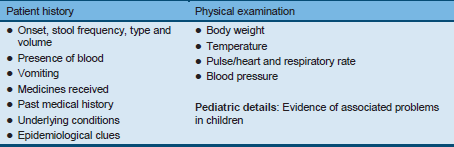
Table 6 Assessment of dehydration using the “Dhaka method”
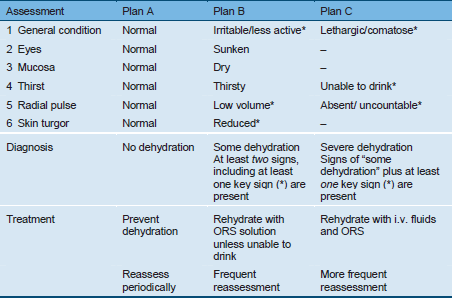
* Key signs.
Laboratory evaluation
For acute enteritis and colitis, maintaining adequate intravascular volume and correcting fluid and electrolyte disturbances take priority over identifying the causative agent. Presence of visible blood in febrile patients generally indicates infection due to invasive pathogens, such as Shigella, Campylobacter jejuni, Salmonella, or Entamoeba histolytica. Stool cultures are usually unnecessary for immune-competent patients who present with watery diarrhea, but may be necessary to identify Vibrio cholerae when there is clinical and/or epidemiological suspicion of cholera, particularly during the early days of outbreaks/epidemics (also to determine antimicrobial susceptibility) and to identify the pathogen causing dysentery.
Epidemiologic clues to infectious diarrhea can be found by evaluating the incubation period, history of recent travel in relation to regional prevalence of different pathogens, unusual food or eating circumstances, professional risks, recent use of antimicrobials, institutionalization, and HIV infection risks.
Stool analysis and culture costs can be reduced by improving the selection and testing of the specimens submitted on the basis of interpreting the case information—such as patient history, clinical aspects, visual stool inspection, and estimated incubation period (Tables 7–9).
Table 7 Patient history details and causes of acute diarrhea
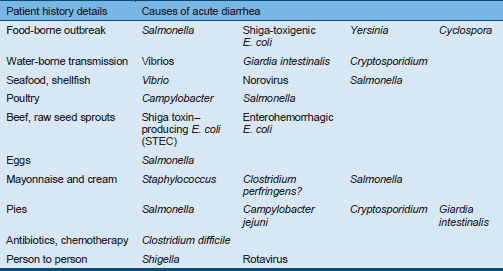
Table 8 Incubation period and likely causes of diarrhea

Table 9 Patient details and bacterial testing to consider

Wherever possible: fecal analysis in cases of severe bloody inflammatory or persistent diarrhea. This is extremely important for developing management protocols during early outbreaks or epidemics.
Screening usually refers to noninvasive fecal tests. Certain laboratory studies may be important when the underlying diagnosis is unclear or diagnoses other than acute gastroenteritis are possible. Where applicable, rapid diagnostic tests (RDTs) may be considered for cholera quick testing at the patient’s bedside.
Pediatric details. Identification of a pathogenic bacterium, virus, or parasite in a stool specimen from a child with diarrhea does not indicate in all cases that it is the cause of illness.
Measurement of serum electrolytes may be required in some children with a longer duration of diarrhea with moderate or severe dehydration, particularly with an atypical clinical history or findings. Hypernatremic dehydration is more common in well-nourished children and those infected with rotavirus, and features irritability, increased thirst disproportionate to clinical dehydration, and a doughy feel to the skin. This requires specific rehydration methods.
Prognostic factors and differential diagnosis in children
Table 10 Prognostic factors in children

Differential diagnosis of acute diarrhea in children:
- Pneumonia—may occur together with diarrhea in developing countries
- Otitis media
- Urinary tract infection
- Bacterial sepsis
- Meningitis
Integrated management of childhood illness (IMCI). In developing countries, a large proportion of childhood morbidity and mortality is caused by five conditions: acute respiratory infections, diarrhea, measles, malaria, and malnutrition. The IMCI strategy has been developed to address the overall health of children presenting with signs and symptoms of more than one condition. In such cases, more than one diagnosis may be necessary and treatments for the conditions may have to be combined. Care needs to be focused on the child as a whole and not just the individual diseases or conditions affecting the child, while other factors that affect the quality of care delivered to children—such as drug availability, organization of the health-care system, referral pathways and services, and community behaviors—are best addressed through an integrated strategy.
The IMCI strategy encompasses a range of interventions to prevent and manage major childhood illness, both in health facilities and in the home. It incorporates many elements of the diarrheal and acute respiratory infection control program, as well as child-related aspects of malaria control, nutrition, immunization, and essential drugs program (WHO, Bangladesh; see www.whoban.org).
4. Treatment Options and Prevention
Rehydration in adults and children
Oral rehydration therapy (ORT) is the administration of appropriate solutions by mouth to prevent or correct diarrheal dehydration. ORT is a cost-effective method of managing acute gastroenteritis and it reduces hospitalization requirements in both developed and developing countries.
Global ORS coverage rates are still less than 50% and efforts must be made to improve coverage.
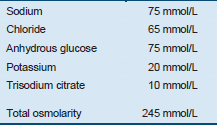
Oral rehydration salts (ORS), used in ORT, contain specific amounts of important salts that are lost in diarrhea stool. The new lower-osmolarity ORS (recommended by WHO and UNICEF) has reduced concentrations of sodium and glucose and is associated with less vomiting, less stool output, lesser chance of hypernatremia, and a reduced need for intravenous infusions in comparison with standard ORS (Table 11). This formulation is recommended irrespective of age and the type of diarrhea including cholera.
ORT consists of:
- Rehydration—water and electrolytes are administered to replace losses.
- Maintenance fluid therapy to take care of ongoing losses once rehydration is achieved (along with appropriate nutrition).
Table 11 Constituents of oral rehydration salts (ORS)
ORT is contraindicated in the initial management of severe dehydration and also in children with paralytic ileus, frequent and persistent vomiting (more than four episodes per hour), and painful oral conditions such as moderate to severe thrush (oral candidiasis). However, nasogastric administration of ORS solution is potentially life-saving when intravenous rehydration is not possible and the patient is being transported to a facility where such therapy can be administered.
Rice-based ORS is superior to standard ORS for adults and children with cholera, and can be used to treat such patients wherever its preparation is convenient. It is not superior to standard ORS in the treatment of children with acute noncholera diarrhea, especially when food is given shortly after rehydration, as is recommended to prevent malnutrition.
Supplemental zinc therapy, multivitamins, and minerals in children
Zinc deficiency is widespread among children in developing countries. Routine zinc therapy, as an adjunct to ORT is useful in modest reduction of the severity but more importantly reduce diarrhea episodes in children in developing countries. The recommendation for all children with diarrhea is 20 mg of zinc per day for 10 days. Infants aged 2 months or younger should receive 10 mg per day for 10 days.
Supplementation with zinc sulfate in recommended doses reduces the incidence of diarrhea during the following 3 months, and reduces nonaccidental deaths by as many as 50%. It is more important in the management of diarrhea in malnourished children and persistent diarrhea. The WHO and UNICEF recommend routine zinc therapy for children with diarrhea, irrespective of the types.
Table 12 Recommended daily allowance (RDA) guide for a 1-year-old child
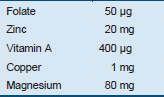
All children with persistent diarrhea should receive supplementary multivitamins and minerals, including magnesium, each day for 2 weeks. Locally available commercial preparations are often suitable; tablets that can be crushed and given with food are least costly. These should provide as broad a range of vitamins and minerals as possible, including at least two recommended daily allowances (RDAs) of folate, vitamin A, zinc, magnesium, and copper (WHO 2005).
Diet
The practice of withholding food for > 4 hours is inappropriate—normal feeding should be continued for those with no signs of dehydration, and food should be started immediately after correction of some (moderate) and severe dehydration, which usually takes 2–4 hours, using ORT or intravenous rehydration.
Pediatric details. Breastfed infants and children should continue receiving food, even during the rehydration phase. However, for non-breastfed, dehydrated children and adults, rehydration is the first priority and that can be accomplished in 2–4 hours.
The notes below apply to both adults and children unless age is specified.
Table 13 Dietary recommendations

Probiotics are live microorganisms, such as Lactobacillus GG (ATCC 53103), with demonstrated beneficial health effects in humans. However, the effects are strain-specific and need to be verified for each strain in human studies. Extrapolation from the results of even closely related strains is not possible, and significantly different effects have been reported. Use of probiotics may not be appropriate in resource-constrained settings, mostly in developing countries.
Pediatric details. Controlled clinical intervention studies and meta-analyses support the use of specific probiotic strains and products in the treatment and prevention of rotavirus diarrhea in infants.
Probiotics for the treatment of acute diarrhea
It has been confirmed that different probiotic strains (see Tables 8 and 9 in WGO's Guideline on probiotics at http://www.worldgastroenterology.org/probiotics-prebiotics.html) including L. reuteri ATCC 55730, L. rhamnosus GG, L. casei DN-114 001, and Saccharomyces cerevisiae (boulardii) are useful in reducing the severity and duration of acute infectious diarrhea in children. The oral administration of probiotics shortens the duration of acute diarrheal illness in children by approximately 1 day.
Several meta-analyses of controlled clinical trials have been published that show consistent results in systematic reviews, suggesting that probiotics are safe and effective. The evidence from studies on viral gastroenteritis is more convincing than the evidence on bacterial or parasitic infections. Mechanisms of action are strain-specific: there is evidence for efficacy of some strains of lactobacilli (e.g., Lactobacillus casei GG and Lactobacillus reuteri ATCC 55730) and for Saccharomyces boulardii. The timing of administration is also of importance.
Prevention of acute diarrhea
In the prevention of adult and childhood diarrhea, there is only suggestive evidence that Lactobacillus GG, L. casei DN-114 001, and S. boulardii are effective in some specific settings (see Tables 8 and 9 in WGO's Guideline on probiotics at http://www.worldgastroenterology.org/probiotics-prebiotics.html).
Antibiotic-associated diarrhea
In antibiotic-associated diarrhea, there is strong evidence of efficacy for S. boulardii or L. rhamnosus GG in adults or children who are receiving antibiotic therapy. One study indicated that L. casei DN-114 001 is effective in hospitalized adult patients for preventing antibiotic-associated diarrhea and C. difficile diarrhea.
Radiation-induced diarrhea
There is inadequate research evidence to be certain that VSL#3 (Lactobacillus casei, L. plantarum, L. acidophilus, L. delbrueckii, Bifidobacterium longum, B. breve, B. infantis, and Streptococcus thermophilus) is effective in the treatment of radiation-induced diarrhea.
Nonspecific antidiarrheal treatment
None of these drugs addresses the underlying causes or effects of diarrhea (loss of water, electrolytes, and nutrients). Antiemetics are usually unnecessary in acute diarrhea management, and some that have sedative effects may make ORT difficult.
Pediatric details. In general, antidiarrheals have no practical benefits for children with acute or persistent diarrhea.
Table 14 Nonspecific antidiarrheal agents

Antimicrobials in adults and children
Table 15 Antimicrobial agents for the treatment of specific causes of diarrhea

Important notes
- All doses shown are for oral administration.
- Selection of an antimicrobial should be based on the susceptibility patterns of strains of the pathogens in the locality/region.
- Antimicrobials are reliably helpful and their routine use is recommended in the treatment of severe (clinically recognizable):
- Cholera, shigellosis, typhoid and paratyphoid fevers.
- Dysenteric presentation of campylobacteriosis and nontyphoidal salmonellosis when they cause persistent diarrhea, and when host immune status is compromised for any reason such as severe malnutrition, chronic liver disease, or lymphoproliferative disorders.
- Invasive intestinal amebiasis.
- Symptomatic giardiasis (anorexia and weight loss, persistent diarrhea, failure to thrive).
- Consider antimicrobial treatment for:
- Shigella, Salmonella, Campylobacter (dysenteric form) or parasitic infections.
- Nontyphoidal salmonellosis among at-risk populations (malnutrition, infants and elderly, immunocompromised patients, and those with liver diseases and lymphoproliferative disorders), and in dysenteric presentation.
- Moderate/severe traveler’s diarrhea or diarrhea with fever and/or with bloody stools.
- Antimicrobials are also indicated for associated health problems such as pneumonia.
- Amebae. Nonpathogenic amebae are more often detected in stool microscopy and get wrongly treated. The presence of ingested erythrocyte in an ameba (hematophagus) stool microscopy indicates invasiveness and a need for treatment; also when the presentation is dysenteric and no other invasive pathogen has been detected. Treatment for amebiasis should ideally include diloxanide furoate following the metronidazole, to get rid of the cysts that may remain after the metronidazole treatment; nitazoxanide is an alternative.
- Azithromycin is widely available and has the convenience of single dosing. For treating most types of common bacterial infection, the recommended azithromycin dosage is 250 mg or 500 mg once daily for 3–5 days. Pediatric dosage: the azithromycin dosage for children can range (depending on body weight) from 10 mg to 20 mg per kilogram of body weight per day, once daily for 3 days.
- Campylobacter. Quinolone-resistant Campylobacter is present in several areas of South-East Asia (e.g., Thailand) and azithromycin is the appropriate option in such situations.
- Cholera. Routine antimicrobial therapy is recommended for treatment of severe (clinically recognizable) cholera. The actual selection of an antimicrobial will depend on recent susceptibility of the pathogen in specific countries; in the absence of such information, susceptibility reports from neighboring countries is the only other choice.
- Erythromycin is hardly used for diarrhea today.
- Nitazoxanide is an effective antiprotozoal in the treatment of diarrhea caused by parasites such as Giardia intestinalis, Entamoeba histolytica, and Cryptosporidium parvum.
- Traveler’s diarrhea. For adults with acute diarrhea, there is good evidence that a single-dose therapy with some newer quinolones, such as ciprofloxacin, shortens the duration of acute traveler’s diarrhea. However, this is still controversial; use should be limited to high-risk individuals or those needing to remain well for short visits to a high-risk area. Antimicrobials should be considered the drugs of choice for empirical treatment of traveler’s diarrhea and of community-acquired secretory diarrhea when the pathogen is known (Table 15).
Pediatric details:
- If drugs are not available in liquid form for use in young children, it may be necessary to use tablets and estimate the doses given in Table 15.
- Consider antimicrobial treatment for:
- When Shigella, Salmonella, Campylobacter (dysenteric form) are the only pathogen isolated from children with persistent diarrhea.
- Nontyphoidal salmonellosis in infants.
- Alternative antimicrobials for treating cholera in children are trimethoprim/sulfamethoxazole (TMP/SMX; 5 mg/kg TMP + 25 mg/kg SMX, 12-hourly for 3 days), and norfloxacin.
Prevention of diarrhea with vaccines
- Salmonella typhi: two typhoid vaccines (with limited cost-efficiency) currently are approved for clinical use.
- Shigella organisms: three vaccines have been shown to be immunogenic and protective in field trials. Parenteral vaccines may be useful for travelers and military personnel, but are impractical for use in developing countries. More promising is a single-dose live-attenuated vaccine currently under development in several laboratories.
- V. cholerae: the current price and need for multiple doses (at least two) and shorter protective efficacy are limitations. A new, cheaper killed-cell vaccine is likely to be available soon; oral cholera vaccines are still being investigated, and their use is recommended only in complex emergencies such as epidemics. Their use in endemic areas remains controversial. In traveler’s diarrhea, oral cholera vaccine is only recommended for those working in refugee or relief camps, since the risk of cholera for the usual traveler is very low.
- Enterotoxigenic E. coli (ETEC) vaccines: the most advanced ETEC vaccine candidate consists of a killed whole-cell formulation plus recombinant cholera toxin B subunit. No vaccines are currently available for protection against Shiga toxin–producing E. coli infection.
Pediatric details:
- Salmonella typhi: no available vaccine is currently suitable for routine use for children in developing countries.
- Rotavirus: in 1998, a rotavirus vaccine, RotaShield (Wyeth), was licensed in the USA for routine immunization of infants. In 1999, production was stopped after the vaccine was causally linked to intussusception in infants. Other rotavirus vaccines are being developed, and preliminary trials are promising. Currently, two vaccines have been approved: a live oral vaccine, RotaTeq, made by Merck for use in children, and GSK’s Rotarix.
- Measles immunization can substantially reduce the incidence and severity of diarrheal diseases. Every infant should be immunized against measles at the recommended age.
5. Clinical Practice
Approach in adults with acute diarrhea
- Perform initial assessment.
- Manage dehydration.
- Prevent dehydration in patients with no signs of dehydration, using home-based fluids or ORS solution.
- Rehydration of patients with some dehydration using ORS, and correct dehydration of a severely dehydrating patient with an appropriate intravenous fluid.
- Maintain hydration using ORS solution.
- Treat symptoms (if necessary, consider bismuth subsalicylate or loperamide in cases of nondysenteric traveler’s diarrhea).
- Stratify subsequent management:
- Epidemiological clues: food, antibiotics, sexual activity, travel, day-care attendance, other illness, outbreaks, season.
- Clinical clues: bloody diarrhea, abdominal pain, dysentery, wasting, fecal inflammation.
- Obtain a fecal specimen for analysis:
- If there is severe, bloody, inflammatory, or persistent diarrhea, and at the beginning of an outbreak/ epidemic.
- Consider antimicrobial therapy for specific pathogens.
- Report to the public health authorities.
- In outbreaks, save culture plates and isolates; freeze fecal specimens and food or water specimens at –70 °C
- Notifiable in the USA: cholera, cryptosporidiosis, giardiasis, salmonellosis, shigellosis, and infection with Shiga toxin–producing E. coli (STEC).
Approach in children with acute diarrhea
In 2002, WHO and UNICEF revised their recommendations for routine use of hypo-osmolar ORS, and in 2004 recommended routine use of zinc as an adjunct to ORT for treatment of childhood diarrhea, irrespective of etiology. Since then, more than 40 countries throughout the world have adopted the recommendations. In countries where both the new ORS and zinc have been introduced, the rate of ORS usage has dramatically increased. Principles of appropriate treatment for children with diarrhea and dehydration:
- No unnecessary laboratory tests or medications.
- Use ORS for rehydration:
- Perform ORT rapidly, within 3–4 hours.
- Routine adjunct zinc therapy for children aged 5 years or younger.
- When dehydration is corrected, rapid re-alimentation:
- Normal food or age-appropriate unrestricted diet.
- Continue breastfeeding.
- Administer additional ORS for ongoing losses through diarrhea.
Table 16 Treatment for children based on the degree of dehydration
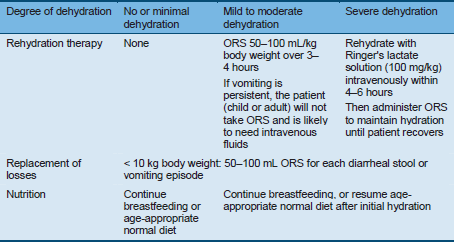
Cautionary notes
- It is dangerous to treat patients with severe diarrheal dehydration using 5% dextrose with 1/4 normal saline, and the risk of death is very high. In diarrheal dehydration, not only water but also a number of electrolytes are lost; the important ones are sodium, potassium, and bicarbonate.
- The loss of sodium is greater in cholera than in ETEC diarrhea (60–110 mmol/L), followed by rotavirus diarrhea (around 20–40 mmol/L)—three leading causes of severe dehydrating diarrhea.
- Efforts to correct dehydration using solutions with lower amounts of sodium (such as 38.5 mmol/L in 1/4 saline with 5% dextrose) would lead to sudden and severe hyponatremia with a high risk of death.
- Ringer’s lactate is the appropriate solution for management of severe dehydration, but normal saline may be life-saving, irrespective of age, when Ringer’s lactate is not available. In such cases, ORS should be initiated as soon as patients (adults and children) are able to drink, to replace bicarbonate and potassium lost in diarrheal stools, particularly for children.
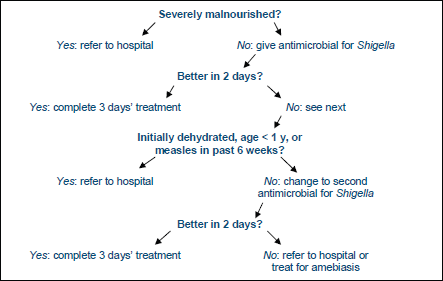
- For acute bloody diarrhea (dysentery) in children, the main principles of the therapeutic approach are:
- Treatment of dehydration
- Stool microscopy to assess the need for antimicrobial therapy. Demonstration of invasive forms of E. histolytica and vegetative Giardia intestinalis in a symptomatic patient would provide a direct diagnosis, and the presence of inflammatory cells would indicate invasive diarrhea and institution of an appropriate antimicrobial agent after a fecal specimen has been sent for culture where possible.
- Frequent, smaller meals with higher protein intakes.
Fig. 1 Therapeutic approach to acute bloody diarrhea in children.
Home management of acute diarrhea in adults and children
Milder and uncomplicated cases of nondysenteric diarrhea in both adults and children can be treated at home, regardless of the etiologic agent, using home-based fluid or ORS as appropriate. Parents/caregivers of children should be educated to recognize signs of dehydration, and when to take children to health facility for treatment. Early intervention and administration of home-based fluids/ORS reduces dehydration, malnutrition, and other complications and leads to fewer clinic visits and potentially fewer hospitalizations and deaths.
Self-medication is safe in otherwise healthy adults. It relieves discomfort and social dysfunction. There is no evidence that it prolongs the illness. However, this may not be appropriate in developing countries where diarrhea requiring specific interventions is more prevalent and people may not be competent in assessing their conditions.
Principles of self-medication:
- Maintain adequate fluid intake.
- Consumption of solid food should be guided by appetite in adults; small, but more frequent meals for children.
- Antidiarrheal medication with loperamide (flexible dose according to loose bowel movements) may diminish diarrhea and shorten the duration.
- Antimicrobial treatment is reserved for prescription only in residents’ diarrhea or for inclusion in travel kits (add loperamide).
Where feasible, families in localities with a high prevalence of diarrheal diseases should be encouraged to store a few ORS packets and zinc tablets if there are children under the age of five in the family for initiating home therapy as soon as diarrhea starts.
Home-made oral fluid recipe
Preparing 1 L of oral fluid using salt, sugar and water at home. The ingredients to be mixed are:
• One level teaspoon of salt.
• Eight level teaspoons of sugar.
• One liter (five cupfuls) of clean drinking water, or water that has been boiled and then cooled.
Antidiarrheal agents. Among hundreds of over-the-counter products promoted as antidiarrheal agents, only loperamide and bismuth subsalicylate have sufficient evidence of efficacy and safety.
Family knowledge: family knowledge about diarrhea must be reinforced in areas such as prevention, nutrition, ORT/ORS use, zinc supplementation, and when and where to seek care.
Indications for medical consultation or in-patient care are:
- Caregiver’s report of signs consistent with dehydration
- Changing mental status
- History of premature birth, chronic medical conditions, or concurrent illness
- Young age (< 6 months or < 8 kg weight)
- Fever ≥ 38 °C for infants < 3 months old or ≥ 39 °C for children aged 3–36 months
- Visible blood in stool
- High-output diarrhea, including frequent and substantial volumes
- Persistent vomiting, severe dehydration, persistent fever
- Suboptimal response to ORT, or inability of caregiver to administer ORT
- No improvement within 48 hours—symptoms exacerbate and overall condition gets worse
- No urine in the previous 12 hours
Cascades
A cascade is a hierarchical set of diagnostic or therapeutic techniques for the same disease, ranked by the resources available. Cascades for acute diarrhea are shown in Figs. 2–4.
Fig. 2 Cascade for acute, severe, watery diarrhea—cholera-like, with severe dehydration. See above for the recipe for home-made oral fluid. ORT, oral rehydration therapy.
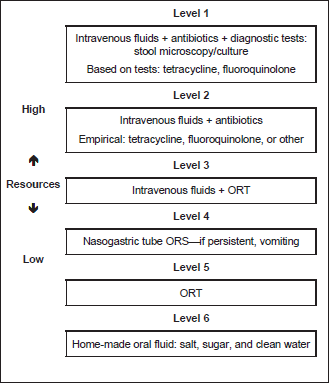
Cautions
- If facilities for referral are available, patients with severe dehydration (at risk of acute renal failure or death) should be referred to the nearest health-care facility with access to intravenous fluids (levels 5 and 6 cannot replace the need for referral in case of severe dehydration).
- Levels 5 and 6 must be seen as interim measures and are better than no treatment if no intravenous facilities are available.
- When intravenous therapy is used, it must be ensured that disposable sterile syringes, needles and drip sets are used, to avoid the risk of hepatitis B and C.
FDA Warning/Regulatory Alert
- August 15, 2013 – Fluoroquinolone Antibacterial Drugs: The U.S. Food and Drug Administration (FDA) has required the drug labels and Medication Guides for all fluoroquinolone antibacterial drugs be updated to better describe the serious side effect of peripheral neuropathy. This serious nerve damage potentially caused by fluoroquinolones may occur soon after these drugs are taken and may be permanent.
Notes
- Nasogastric therapy requires skilled staff.
- Often, intravenous fluid treatment is more easily available than nasogastric tube feeding. (Caution: there is a risk of infection with contaminated intravenous infusion equipment.)
Pediatric details
- Nasogastric feeding is not very feasible for healthy and active older children, but it is suitable for malnourished, lethargic children.
- Nasogastric administration (ORS and diet) is especially helpful in long-term severely malnourished children (anorexia).
Fig. 3 Cascade for acute, mild/moderate, watery diarrhea—with mild/moderate dehydration. See above for the recipe for home-made oral fluid. ORT, oral rehydration therapy.

Fig. 4 Cascade for acute bloody diarrhea—with mild/moderate dehydration. See above for the recipe for home-made oral fluid. ORT, oral rehydration therapy.

























