Julio C. Bai (Chair, Argentina), Carolina Ciacci (Co-chair, Italy), Gino Roberto Corazza (Italy), Michael Fried (Switzerland), Carolina Olano (Uruguay), Mohammad Rostami-Nejad (Iran), Andrea González (Argentina), Peter Green (USA), Javier Gutierrez-Achury (UK/Netherlands), Michael Schultz (New Zealand), Elena Verdú (Canada), Kassem Barada (Lebanon), Peter Gibson (Australia), Sibylle Koletzko (Germany), Thierry Coton (France), Chris Mulder (Netherlands), Govind Makharia (India), Anton LeMair (Netherlands)
1. Definitions
Gluten can be defined as the rubbery protein mass that remains when wheat dough is washed to remove starch [1]. The major protein components of gluten, gliadins and glutenins, are storage proteins in wheat. Gluten and gluten-related proteins are present in wheat, rye, and barley and are used widely in food processing to give dough the desired baking properties, add flavors, and improve texture. Gluten exposure in genetically predisposed individuals may lead celiac disease [2,3].
1.1 Celiac disease
Celiac disease (CD) is a chronic, multiple-organ autoimmune disease that affects the small intestine in genetically predisposed children and adults. It is precipitated by the ingestion of gluten-containing foods [3]. It is also referred to as celiac sprue, gluten-sensitive enteropathy, or nontropical sprue.
1.2 Wheat allergy
Wheat allergy is an adverse immunologic reaction, mediated by immunoglobulin E (IgE) and non-IgE, to different wheat proteins. Depending on the route of allergen exposure and the underlying immunologic mechanisms, wheat allergy can be classified into four main categories [4]:
- Classic food allergy, affecting the skin, gastrointestinal or respiratory tract
- Exercise-induced anaphylaxis
- Occupational asthma (baker’s asthma) and rhinitis
- Contact urticaria
1.3 Nonceliac gluten sensitivity
Patients who experience resolution of both gastrointestinal and nongastrointestinal symptoms with a gluten-free diet (GFD), when celiac disease as well as IgE-mediated wheat allergy (WA) have been excluded, are labeled as having “nonceliac gluten sensitivity” (NCGS) [5]. Despite the often self-reported substantial improvement of symptoms that patients experience with a GFD, the results in clinical trials (gluten versus placebo) have so far been conflicting [6–10], leading to uncertainty about the true frequency of such a syndrome.
At the moment, the only way to detect specific food sensitivity (when a specific mechanical test is not available) is to carry out double-blind placebo-controlled cross-over food challenges using wheat protein minimally contaminated with other inducers of symptoms, such as fructosans. However, nocebo effects, a normal phenomenon, are present in such challenges and need to be taken into consideration when the results are being interpreted [8,9]. Other potential or concomitant alimentary factors that are able to induce symptoms include carbohydrates (fermentable oligo-, di-, monosaccharides and polyols, FODMAPs) and wheat proteins such as amylase/trypsin inhibitors (ATIs). In addition, as there is still no concrete proof that gluten is responsible for the symptoms or the precise mechanism involved, some authors believe that “nonceliac wheat hypersensitivity” or “wheat intolerance syndrome” would be more appropriate terms [11,12].
2. Cascades and key points in diagnosis management
2.1 WGO Cascades
The World Gastroenterology Organisation (WGO) aims to guide health providers in the best management of celiac disease and other gluten-related disorders (GRDs) by providing a concise document with recommendations based on the latest evidence and resulting from our global expert consensus process, based on best current practices.
In their diagnostic and treatment cascades, WGO guidelines provide a resource-sensitive and context-sensitive approach. A standardized global approach would require the epidemiology and clinical setting to be comparable in all parts of the world, with the full ranges of diagnostic tests and medical treatment options also generally available. This is not the case: neither the epidemiology nor the availability of resources for the diagnosis and management of celiac disease and GRDs is sufficiently uniform throughout the world to support the provision of a single “gold standard” approach. This Global WGO Guideline, therefore, includes a set of cascades to provide context-sensitive and resource-sensitive options for the diagnosis and management of GRDs. As such, the WGO Cascades are intended to serve as a “global” complement to, rather than a replacement for, the “gold standard” guidelines produced by regional groups and national societies. The Appendix here lists current gold standard guidelines.
As there is a lack of sufficient data on NCGS and wheat allergy, the authors of this guideline consider that it is not at present feasible to produce cascades for NCGS and wheat allergy.
WGO cascades: a hierarchical set of diagnostic, therapeutic, and management options to deal with risk and disease, ranked by the resources available.
WGO guidelines and cascades are intended to highlight appropriate, context-sensitive and resource-sensitive management options for all geographical areas, regardless of whether they are considered to be “developing,” “semi-developed,” or “developed.” WGO cascades are context-sensitive, and the context is not necessarily defined solely by resource availability.
2.2 Cascade for diagnosing celiac disease
A diagnosis based only on “clinical assessment” and improvement after a gluten-free diet should be strongly discouraged. This has been a source of misdiagnosis and can only be helpful in a minority of patients out of the overall population (those with classic celiac disease who have signs and symptoms of malabsorption) and in areas with extremely limited resources. It could cause confusion, leading to incorrect diagnosis of celiac disease in patients with nonceliac disease gluten sensitivity, non–gluten-related enteropathy, irritable bowel syndrome, or “by chance” improvement of symptoms (e.g., after intestinal infection). In fact, a gluten-free diet can produce a nonspecific effect due to non–gluten-dependent dietary modifications, lower levels of FODMAPs, or because of a “placebo effect.”
As first-line tests for symptomatic and asymptomatic patients, the experts suggest anti-tissue transglutaminase (anti-tTG) IgA + total IgA. However, to confirm a positive first test, or in case of borderline tTG IgA titers, it is recommended that other tests should be added. Other immunoassays regarded as good tests include anti-endomysium antibody (EMA) and either a positive IgA-transglutaminase 2 (TG2) test or positive IgG-deamidated gliadin peptide (DGP) test. The combination of IgG-DGP and IgA-TG2 is particularly useful as an addition, to detect patients with CD who are IgA-deficient. IgG-DGP is reported to be able to detect a few more IgA-sufficient patients who are missed on IgA-TG2 tests. For some authors, performing IgA-tTG + IgG-DGP — two tests addressing different antigens — is better than if two tests addressing the same autoantigen (IgA-tTG + IgA-EMA) are carried out.
Table 1 Cascade with resource-sensitive options for diagnosing celiac disease
Resource level
|
Cascade of diagnostic options
|
|
Gold standard
|
Medical history and physical examination
|
|
Celiac disease–specific antibodies: assessment and intestinal biopsy
|
- Anti-tTG IgA or anti-EMA IgA, and total IgA to exclude IgA deficiency
|
- In case of selective IgA deficiency, IgG-based tests should be used: anti-DGP, anti-tTG, or EMA (the latter two are highly sensitive, but with lower specificity)
|
- Intestinal biopsies are always necessary if the antibodies are low or negative, and if there are no signs/symptoms of malabsorption. However, pitfalls in histological diagnosis are common, and findings are characteristic, but not specific
|
- In certain situations, biopsies may be omitted after the pros and cons have been discussed with an expert physician with special knowledge of celiac disease. The ESPGHAN has suggested a very high tTG-IgA titer (> 10 times the upper limit of normal), positive EMA in a second blood sample, signs of malabsorption
|
|
Medium resources
|
Medical history and physical examination
|
|
Antibody assessment as a single diagnostic tool — when endoscopy is not possible or trained pathologists are not available; titer levels should be considered
|
- IgA anti-tTG and total IgA are the most appropriate and cost-effective approach in patients with suspected celiac disease
|
- In IgA-deficient patients, an IgG-based test (anti-DGP, anti-tTG, EMA) is recommended
|
- In patients with a positive serological anti-tTG test result in a setting in which histology is not possible, the likelihood of celiac disease can be further increased with a confirmatory test (anti-DGP, EMA) in a second blood sample
|
- One should be aware of possible low-titer, false-positive anti-tTG in patients with thyroid or liver disease
|
|
Intestinal biopsies as a single tool,* in settings in which pathology is (perhaps remotely) available but clinical laboratories cannot achieve the required standards
|
- Pitfalls in histological diagnosis are common and should be considered when nonexpert pathologists are assessing biopsies. Findings are characteristic, but not specific
|
- The strategy should be combined with the demonstration of clinical improvement after introduction of a gluten-free diet
|
- A second biopsy may be necessary if there is no clinical improvement
|
- A later gluten challenge to confirm biopsies should be performed, particularly if the diagnosis was established in young children, in whom other types of enteropathy are common
|
|
Low resources
|
Medical history and physical examination
|
|
Antibody assessment as a single diagnostic tool
|
- Start with testing anti-tTG IgA
|
- If the results are negative and there is still a suspicion of celiac disease, add total IgA or DGP-IgG, if available
|
Notes
* Regional differences may be important — for example, when considering tropical sprue.
- The performance of IgG and IgA anti-DGP is similar to that of IgA anti-tTG in symptomatic patients.
- Anti-tTG IgA is more sensitive, but less specific than EMA IgA.
- A high antibody titer is more likely to be diagnostic.
Anti-tTG, antibodies to tissue transglutaminase-2; EMA IgA, endomysial antibodies IgA; anti-DGP, antibodies to deaminated gliadin peptides.
2.3 Cascade for managing celiac disease
Resource level
|
Cascade of options
|
|
Gold standard
|
Clinical evaluation (including lab tests) + dietary interview by dietitian
|
|
|
Serology tests and intestinal mucosal biopsies
|
|
|
Dietary instructions — lifelong and strict gluten-free diet —regular dietetic follow-up
|
|
|
- Facilitate the patient’s approach to support groups for celiac patients with acknowledged experience
|
|
|
- Follow-up* monitoring, including antibody tests: after 3–6 months in the first year and once a year thereafter in stable patients responding to the gluten-free diet
|
|
|
- The role of repeat biopsies in the follow-up period is still controversial, and they are not universally recommended. Repeat biopsies should be considered mandatory for seronegative patients. Repeat biopsies are recommended in patients with persistent symptoms during a strict gluten-free diet
|
|
Medium resources
|
Clinical evaluation (including lab tests) + dietary interview by expert dietitian, if available
|
|
|
Serology tests — anti-tTG IgA and DGP IgA to monitor mucosal damage and consumption of gluten
|
|
|
Dietary instructions— lifelong and strict gluten-free diet — regular dietetic follow up
|
|
|
- Facilitate the patient’s approach to support groups for celiac disease patients with acknowledged experience
|
|
|
- Follow-up* monitoring: on demand in first year
|
|
|
- Duodenal biopsies after a gluten challenge should be considered for cases that have negative serology at diagnosis to confirm celiac disease
|
|
|
- Biopsies may be also considered for patients with persistent symptoms
|
|
Low resources
|
Clinical evaluation (including lab tests if possible) + dietary interview by physician or expert dietitian, if available
|
|
|
Dietary instructions: lifelong and strict gluten-free diet — dietetic follow-up when possible
|
|
|
- Facilitate the approach to support groups for celiac patients with acknowledged experience
|
|
|
- Follow-up* monitoring: on demand and symptom-based
|
Anti-tTG, antibodies to tissue transglutaminase; DGP, deaminated gliadin peptides; IgA, immunoglobulin A.
* Follow-up:
- The most important obstacle to implementing the recommendations is poor awareness of celiac disease by patients and physicians.
- Among gastroenterologists, there is a sense that celiac disease does not require follow-up by a specialized physician after diagnosis and may be considered a minor condition in comparison with irritable bowel disease (IBD) and inflammatory bowel syndrome (IBS).
2.4 Key points
- Gluten and gluten-related proteins present in wheat, rye, barley, and in oats when contamination with wheat has not been avoided, are the causative external trigger of celiac disease. Celiac disease occurs almost exclusively in patients who express the major histocompatibility complex (MHC) class II human leukocyte antigen, DQ subregion 2 (HLA-DQ2) and HLA-DQ8 molecules.
- The incidence has been increasing over the last 20 years.
- The prevalence of celiac disease in the population varies between roughly one in 100 and one in 300 in most parts of the world.
- The highest incidence of celiac disease seroconversion is between 12 and 36 months of age.
First-degree and (to a lesser extent) second-degree relatives have an increased risk for celiac disease (approximately 70% of first-degree relatives are HLA-DQ2–positive and/or HLA-DQ8–positive, in comparison with 30–35% in the general population). The clinical presentation of celiac disease varies widely, and the onset of the disease or symptoms may occur at any time in life. Many patients with celiac disease have few gastrointestinal symptoms or present with extraintestinal manifestations, while a minority of patients have malabsorption (classic celiac disease). Patients with symptomatic celiac disease appear to have an increased risk of complications, including death, in comparison with the general population. However, this excess rate of major complications appears to resolve after 3–5 years on a strictly gluten-free diet. The risk for asymptomatic celiac disease cases that are only detected on serological screening is poorly known.
Key diagnostic findings include:
- Characteristic histopathologic changes in intestinal mucosal biopsies, including intraepithelial lymphocytosis, crypt hyperplasia, and various grades of villous atrophy.
- Evidence that small-intestinal enteropathy is dependent on gluten, which can be shown by positive celiac disease–specific antibodies and/or clinical and/or histological improvement in response to a gluten-free diet.
Serological testing can:
- Confirm celiac disease in patients with characteristic enteropathy
- Identify patients in whom biopsies are warranted
- Investigate patients with an increased risk for the disease
- Help diagnose celiac disease in some suspected cases
Treatment:
Patients with celiac disease should not eat products containing wheat, rye, or barley. Patients usually need to follow a strictly gluten-free diet for the rest of their lives. Oats may be consumed, but they may be contaminated by wheat, and pure, uncontaminated, gluten-free oats are often not available. A small subgroup of patients with celiac disease (fewer than 5%) may also be intolerant to pure oats, and an oat-free diet is therefore not recommended, at least during the first 3 months of treatment.
3. Epidemiology
Celiac disease is common throughout the world, and its prevalence has significantly increased over the past 20 years [13]. There has been a substantial increase in the numbers of new cases of celiac disease, partly due to better diagnostic tools and thorough screening of individuals considered to be at high risk for the disorder [14]. Celiac disease still represents a statistical iceberg, with far more undiagnosed than diagnosed cases [15–17].
- The prevalence in Western countries is around 1% of the general population, but it is substantially increasing in other parts of the world.
- The female-to-male ratio has been variably reported, with prospective population studies rating from 1 : 3 to 1.5 : 1.
- Celiac disease is now known to affect all age groups, including the elderly; more than 70% of new patients are diagnosed above the age of 20 years [18].
- The risk of having celiac disease is much greater in first-degree relatives (up to 10%) and lesser in second-degree relatives, as well in individuals with type 1 diabetes mellitus and other autoimmune diseases, Down syndrome, and a number of other associated diseases [16].
- Clinically severe manifestations may occur during pregnancy or during the puerperium in up to 17% of female patients [19].
3.1 Global emergence of celiac disease
Early epidemiological studies regarded celiac disease as a disease of individuals of Caucasian ancestry, located mainly in Europe and North America [20]. However, further studies in other areas of the world revealed a similar prevalence of celiac disease there [21–23]. The prevalence of the disorder is globally 1%, but wide variations between countries have been shown [17].
- The prevalence of celiac disease in low-risk populations in the developing world is 0.14–5.7%, while that in high risk populations is 1.2–55% [24].
- Some of these studies have detected celiac disease among people with Native American or Afro-American ancestry [25].
- Recent reports have shown that celiac disease is a common disorder in North Africa [26], the Middle East [17], India [27], and Pakistan [28].
- In intertropical Africa, celiac disease has been reported especially in Djibouti and Sudan [29–31]. A low level of awareness, lack of diagnostic tools, and lower gluten exposure due to different diets might explain the apparently lower incidence of celiac disease in sub-Saharan Africa. No local seroprevalence data are available.
- Reports from China have shown that both the celiac disease–predisposing HLA-DQ alleles and celiac disease are not rare in the provinces of Jiangsu and Zhejiang, at least [32].
In summary, the worldwide distribution of gluten-containing foods, predisposing genotypes, and factors involved in the pathogenesis of celiac disease are likely to be responsible for the widespread and almost universal emergence of the disorder.
- A multicenter study in Europe has confirmed the 1% prevalence, with some variability among the different countries.
- A meta-analysis of studies from Asia suggests that celiac disease is not uncommon in Asia and that the seroprevalence and prevalence of celiac disease in Asia are 1.6% and 0.5%, respectively [33].
- A recent epidemiologic study in Amerindians from the north of Argentina (Toba community) attending a multidisciplinary health mission detected celiac disease (2.2%) or persistent autoimmunity (2%) in individuals with the HLA-DQ8 predisposing haplotype [34].
- The highest celiac disease prevalence in the world has been identified in a specific Western Saharan population, at 5.6% — almost ten times higher than in most European countries. The population also showed one of the highest frequencies of HLA-DQ2 and HLA-DQ8 and very high gluten consumption [35].
- Other studies have demonstrated that the number of new cases of celiac disease found in a specific period in a given population (the incidence) is increasing (in North America and Europe) [13,36].
Table 3 Prevalence of celiac disease in different parts of the world
|
|
Country
|
Prevalence
|
Remarks
|
|
Africa
|
Burkina Faso
|
0
|
Wheat consumption is negligible
|
|
Libya
|
0.8%
|
|
|
Sahara
|
5.6%
|
High level of susceptibility genotypes, HLA-DQ2 and HLA-DQ8, very high gluten consumption
|
|
Sub-Saharan Africa
|
n/a
|
Short series in Djibouti, Sudan. Real incidence not known: lack of awareness and diagnostic capabilities, and staple cereals most gluten-free and HLA-related predisposing genes much less frequent than in Western countries
|
|
Tunisia
|
1 : 18–1 : 335
|
|
|
Zambia
|
1 : 18–1 : 335
|
|
|
Americas
|
Argentina
|
1 : 67–1 : 681
|
Toba community
High level of HLA-DQ8 susceptibility genotype
|
|
Brazil
|
1.66 : 1000
|
|
|
Canada
|
0.9%
|
|
|
Chile
|
1 : 67–1 : 681
|
|
|
Colombia
|
1 : 67–1 : 681
|
|
|
Mexico
|
1 : 67–1 : 681
|
|
|
USA
|
1 : 100–200
|
|
|
Europe
|
Belarus
|
n/a
|
|
|
Bulgaria
|
2.65%
|
IDDM patients
|
|
Croatia
|
1 : 519
|
|
|
Czech Republic
|
1 : 218
|
|
|
Denmark
|
6.9 : 100,000
|
|
|
Estonia
|
0.34%
|
|
|
Finland
|
2%
|
|
|
France
|
1 : 250
|
|
|
Germany
|
0.9%
|
|
|
Greece
|
0.18%
8.6%
|
Adults
IDDM children
|
|
Hungary
|
1 : 166
|
|
|
Ireland
|
1 : 300
|
|
|
Italy
|
1 : 106
|
|
|
Latvia
|
0.35–0.49%
|
|
|
Netherlands
|
1 : 198
|
|
|
Norway
|
1 : 262
|
Low wheat consumption, low frequency of HLA-DQ2
|
|
Poland
|
1 : 404
|
In children
|
|
Portugal
|
1 : 134
|
|
|
Romania
|
3.9%
|
IDDM patients
|
|
Russia
|
n/a
|
|
|
Spain
|
1 : 118
|
|
|
Sweden
|
1 : 190
|
|
|
Switzerland
|
1 : 132
|
|
|
UK
|
1 : 100
|
|
|
Ukraine
|
n/a
|
|
|
Middle East
|
Egypt
|
0.5%
|
|
|
Iran
|
1 : 100
|
Several studies have reported prevalences in Iran and other Middle Eastern countries similar to those in western Europe
|
|
|
|
|
|
Kuwait
|
1 : 18
|
Chronic diarrhea
|
|
Turkey
|
1 : 87
|
Anatolian adults: 1 : 100
|
|
Saudi Arabia
|
12 : 100
|
Type 1 diabetes
|
|
Syria
|
1.5 : 100
|
|
|
Yemen
|
1 : 18
|
Chronic diarrhea
|
|
Asia, South-East Asia, Australasia
|
Australia
|
1 : 82–1 : 125
|
|
|
India
|
0.3–1.04%
|
General population; prevalence higher in northern India
|
|
Japan
|
1 : 20.000
|
Low wheat consumption, low frequency of HLA-DQ2
|
|
Indonesia, Korea, Malaysia, Singapore, smaller Pacific Islands
|
n/a
|
|
|
Kazakhstan
|
n/a
|
|
IDDM, insulin-dependent diabetes mellitus; n/a = not available.
The ratio of diagnosed to undiagnosed cases of celiac disease varies from country to country (one to two in Finland, one to 10 in the United States, Argentina, and Germany) [15,21,37,38]. This suggests that most cases of celiac disease would remain undetected without active screening.
There is an urgent need to increase awareness among primary-care physicians and pediatricians about the wide diversity of clinical manifestations and the role of serological testing in the diagnosis of celiac disease. This has been demonstrated in Northern Ireland and Finland as a major factor in the increase in diagnosis in those countries [39,40].
There are no substantial differences between the proportions of symptomatic patients and screening-detected patients (mostly with a subclinical phenotype) in any of the countries or geographic areas in which epidemiological studies have been carried out.
3.2 Genetics
The specific role of the HLA-DQA1 and HLA-DQB1 genes in the presentation of gluten peptides as antigens makes the MHC HLA locus the most important genetic factor in the development of celiac disease. Celiac disease is associated with HLA-DQ2 and -DQ8; HLA-DQ2 is present in approximately 95% of celiac disease patients of northern European descent, while the rest are carriers of HLA-DQ8 [41]. Recent analyses have shown that HLA class I molecules are also associated with celiac disease [42,43].
The frequency of the high-risk HLA-DQ haplotypes for celiac disease shows variations across populations [42], and this may be related to differences in the prevalence of celiac disease — there are marked differences between the HLA risk groups, with an up to six times higher risk in high-risk groups (HLA haplotype DR3-DQ2, especially homozygotes) in comparison with low-risk groups (DR4-DQ8) [44].
- The presence of HLA risk alleles is a necessary, but not a sufficient, factor for the development of celiac disease (< 1% of patients lack DQ2 and DQ8) [45,46].
- Research suggests that, although key to the pathogenesis of celiac disease, HLA haplotypes alone confer approximately 35–40% of the genetic risk [42,47].
- HLA typing tests have a high negative predictive value, and an absence of HLA-DQ2/-DQ8 excludes the presence of celiac disease in susceptible individuals [44,47].
- Additional non-HLA genomic regions identified as being associated with celiac disease appear to explain an additional 32% of the genetic heritability (25.5% for MHC HLA and 6.5% for non-HLA region, calculated in the liability scale using 1% of prevalence) [42].
- In general terms, the presence of gluten in the diet is a necessary factor; there is no celiac disease without gluten, even in the presence of a genetic predisposition.
HLA tests are recommended in the following situations:
- A negative HLA test is helpful to exclude the possibility of celiac disease.
- In individuals with an uncertain diagnosis of celiac disease — negative serology, but histology suggestive of celiac disease. HLA testing is helpful here only if it is negative. HLA testing will most likely be positive if the serology is positive and the histological findings are negative.
- In order to distinguish siblings who can be reassured that it is unlikely that they will develop celiac disease from those who need to be monitored for the development of the disease.
- In individuals with other autoimmune diseases and some genetic disorders who should be investigated for celiac disease (e.g., autoimmune thyroiditis).
3.3 Environmental factors
- Gluten is essential for the development of celiac disease.
- Randomized controlled trials have not confirmed, as was formerly supposed, that early introduction of gluten (< 5 months of age) or later (at 12 months of age) can reduce the risk of celiac disease [48–50]. Whether or not the amount of gluten at an early age matters is still a matter of controversy [51]. The duration of breastfeeding (exclusive, total, or at the time of gluten introduction) is also not associated with a reduced risk of developing celiac disease [48–50,52].
- Loss of gluten tolerance can occur at any time in life as a consequence of other triggers besides gluten.
4. Clinical history and key symptoms
The recent increase in the numbers of patients diagnosed with celiac disease correlates with greater awareness among physicians of the remarkably wide and varied clinical manifestations of this disorder [3,35,38,53,54]. It is also related to the development and regional availability of accurate screening tests, as well as a genuine increase [35,36] in the incidence.
A wide range of symptoms are observed in the clinical setting. Here we follow the “Oslo definitions” for celiac disease [3].
4.1 Classic celiac disease
Patients with classic celiac disease present with signs and symptoms of malabsorption, including diarrhea, steatorrhea, iron-deficiency anemia, and weight loss or growth failure [3].
Adults — signs and symptoms
- Chronic diarrhea (formerly considered the most common symptom)
- Weight loss
- Iron-deficiency anemia
- Abdominal distension due to bloating
- Malaise and fatigue
- Edema (hypoproteinemia)
- Osteoporosis
Children — signs and symptoms
- Failure to thrive, weight loss, growth failure
- Vomiting
- Chronic diarrhea
- Abdominal distension due to bloating
- Iron-deficiency anemia
- Muscle wasting
- Edema (hypoproteinemia).
- Irritability and unhappiness
4.2 Nonclassic celiac disease
In nonclassic celiac disease, patients may present with nonspecific gastrointestinal symptoms without signs of malabsorption, or with extraintestinal manifestations (without gastrointestinal symptoms) [3,55]. The prevalence of nonclassic celiac disease is high in the Western world, with patients mainly presenting with unexplained iron-deficiency anemia.
Adults and children – signs and symptoms
The presentation may be monosymptomatic or oligosymptomatic, or with low intensity.
- Abdominal distension
- Abdominal pain
- Celiac crisis (cholera-like syndrome) [56]
- Chronic fatigue, lack of energy
- Chronic constipation in children
- Chronic migraine
- Dermatological manifestations (such as rash, psoriasis, blisters)
- Peripheral neuropathy
- Unexplained chronic hypertransaminasemia [57]
- Folic acid deficiency
- Reduced bone density
- Unexplained infertility
- Delayed puberty, late menarche/early menopause
- Unexplained miscarriage, premature birth, or small for gestational age infant
- Dental enamel defects
- Dyspepsia, early satiety, anorexia
- Depression and anxiety, moodiness and irritability
- Vitamin B12 deficiency
4.3 Asymptomatic celiac disease
Many patients, especially those diagnosed during screening, report no symptoms even on detailed questioning, and despite the presence of characteristic intestinal lesions. A gluten-free diet often leads to improvement in the quality of life, even in patients who considered themselves asymptomatic at the time of diagnosis [58]. This supports continuation of dietary restriction over the long term [59].
This diversity of symptoms represents a challenge for health professionals who are not familiar with celiac disease.
Family studies have shown that almost 50% of newly diagnosed celiac disease patients have an asymptomatic clinical course [60]. It is therefore likely that half of the undiagnosed population has this asymptomatic clinical form. However, many patients with “asymptomatic” disease report a “new normality” after starting a gluten-free diet, and most of them remain on the diet [61].
5. Diagnosis of celiac disease
5.1 Why is celiac disease difficult to diagnose?
- Wide spectrum of clinical characteristics, which can vary during a patient’s lifetime.
- Symptoms may overlap with those of irritable bowel syndrome.
- Patients may have mainly extraintestinal manifestations, or even no symptoms.
- There is a lack of awareness about celiac disease among clinicians (and patients).
- There is a lack of diagnostic capabilities in some geographical areas, such as intertropical Africa.
There are also several “myths,” such as:
- Celiac disease is rare.
- Celiac disease occurs:
— Only in Caucasians
— Mostly in Europe and the United States.
— Only in childhood
— Only in patients with chronic diarrhea
- Celiac disease can be cured after (a period of) treatment.
- Celiac disease is a mild disorder and “the problem goes away after diagnosis and a gluten-free diet.”
In adults and children, the diagnosis of celiac disease hinges on the presence of positive celiac disease–specific autoantibodies and concomitant diagnostic intestinal biopsies [15].
In symptomatic patients with signs of malabsorption (classic celiac disease), high-titer (10 ×) autoantibodies (anti-transglutaminase) confirmed by positive or other autoantibody tests (EMA or DGPs) in a second blood sample has been suggested as sufficient to diagnose celiac disease, without intestinal biopsies. This should be considered after the pros and cons have been discussed with an expert physician [62].
- For diagnosis, second (post-treatment) biopsies are not necessary for most patients if they have positive initial diagnostic serology findings and respond satisfactorily to a gluten-free diet. There is no wide consensus among experts or guidelines with respect to this statement [63,64].
- The second biopsies should be reserved for patients in whom the first biopsies and serologic tests have been inconclusive (e.g., seronegative enteropathy) or for patients who fail to respond despite adherence to a strict gluten-free diet [65].
- A gluten challenge, in which pure gluten or gluten-containing food is reintroduced, should be reserved for patients who have been started on a gluten-free diet but have a doubtful diagnosis [66,67].
Table 4 Algorithm for diagnosing celiac disease
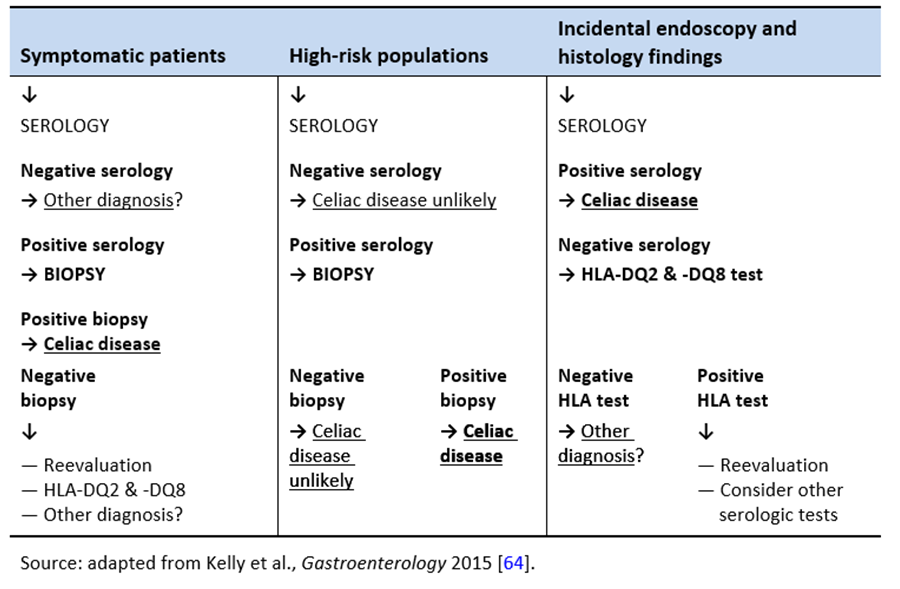
5.2 Which patients should be tested?
A recent review [68] suggests that, although celiac disease fulfills several WHO criteria for mass screening, sufficient proof of its benefit is still lacking and a strategy of proactive case-finding in high-risk groups is the appropriate approach at present.
5.2.1 Patients with clinical manifestations suggestive of celiac disease
- Chronic or intermittent diarrhea (e.g., IBS)
- Iron-deficiency anemia
- Failure to thrive
- Weight loss
- Growth failure
- Delayed puberty
- Primary or secondary amenorrhea
- Persistent fatigue
- Dermatitis herpetiformis–like rash
- Fracture with inadequate trauma/osteopenia/osteoporosis
- Infertility
- Ataxia
- Unexplained increase in transaminases
5.2.2 Conditions associated with an increased risk for celiac disease
- Type 1 diabetes mellitus
- Autoimmune thyroid disease
- Autoimmune liver disease
- Down syndrome
- Turner syndrome
- Williams syndrome
- Selective immunoglobulin A (IgA) deficiency
- Unexplained elevated serum aminotransferase levels
Note: These conditions may not be related pathogenically to celiac disease, but the high prevalence of cases makes it necessary to test patients serologically.
5.2.3 First-degree relatives of patients with celiac disease
- All first-degree relatives should be screened for celiac disease.
- Approximately 7–10% of first-degree relatives may develop celiac disease, and the risk varies considerably along with their relationship to the index patient.
- The risk of having celiac disease is highest in siblings (9%), followed by offspring (8%) and parents (3%). While sisters and daughters of the index patient have the highest risk of having celiac disease (one in seven and one in eight, respectively), the risk is one in 13 in sons, one in 16 in brothers, one in 32 in mothers, and one in 33 in fathers. The risk of having celiac disease is higher among second-degree relatives (2.3%) of patients in comparison with the general population [69].
- HLA typing for all first-degree relatives; do no further work-up on those who are negative for HLA-DQ2/8. The risk for HLA-DQ2/-DQ8–positives varies depending on the alleles present, with the highest risk in homozygotes for DQ2. Seroconversion towards positive antibodies has been estimated at below 1% cumulatively per year, with the highest incidence rate between 12 and 36 months [44].
5.3 Differences in diagnostic criteria for celiac disease
The diagnostic criteria for celiac disease may differ in some parts of the world because of several factors.
- Cut-off titers for most of the serological tests are based on data from Caucasian populations. The cut-off titer for a positive test, sensitivity, and specificity may vary with age, ethnicity, and region of residence.
- While celiac disease is the predominant cause of villous abnormalities in Caucasian populations, in many countries tropical sprue, parasitic infections, and immunoproliferative small-intestinal diseases are also relevant causes of villous abnormalities.
5.4 Diagnostic tests
5.4.1 Endoscopy in patients with suspected celiac disease
Although endoscopy is neither sufficiently sensitive nor specific [70] to detect celiac disease, the following findings [71] on endoscopy should raise a suspicion:
- Scalloping of duodenal folds
- Fissuring over the folds and a mosaic pattern in the mucosa
- Flattening of folds
- Decreased number of folds, size and/or disappearance of folds with maximum insufflation
- Absence of villi at magnification
- Granular appearance of the duodenal bulb
Intestinal mucosal biopsies should always be obtained when any of the above endoscopic features are observed. Endoscopic biopsies should also be obtained even if the endoscopic folds appear normal, but there is a clinical suspicion of the disease, since many patients with celiac disease may have apparently normal folds. An absence of endoscopic findings has a low predictive value for discarding the possibility of celiac disease in low-risk populations [64,70].
5.4.2 Intestinal biopsy and histology
A combination of villous abnormalities seen at intestinal biopsies, together with a positive serological test, is the gold standard diagnostic criterion for celiac disease. A modified Marsh classification for villous abnormalities is now widely used for assessing the severity of villous atrophy in clinical practice [63,72,73].
While the histological changes seen in celiac disease are considered to be characteristic, they are not pathognomonic, as similar changes can be seen in several other conditions, including tropical sprue, parasitic infection, common variable immunodeficiency, human immunodeficiency virus (HIV) enteropathy, and drug and enteropathy induced by food allergy (such as to cow’s milk).
Celiac disease predominantly affects the mucosa of the proximal small intestine, with damage gradually decreasing in severity towards the distal small intestine, although in severe cases the lesions can extend to more distal areas [20].
Lesions in the duodenum/upper jejunum may be patchy, as a result of which they may be missed if sufficient sampling of mucosa has not been done. Four to six biopsy samples [3] must be taken from the second part of the duodenum, and from the duodenal bulb. In patients with ultrashort celiac disease, pathology may be confined to the duodenal bulb — highlighting the importance of taking biopsies from that area [74]. Biopsies must be taken when patients are on a gluten-containing diet (at least 3 g of gluten/day for 2 weeks) [75]. A negative histological diagnosis may justify a second biopsy in selected patients who have positive antibodies, such as high-titer anti-tTG, anti-DGP and/or endomysial antibodies.
Biopsy samples taken from the proximal duodenum above the ampulla of Vater may have artifacts (e.g., stretching of villi) produced by submucosal Brunner’s glands, which may be falsely interpreted as flat mucosa.
Under light microscopy, the most characteristic histological findings [20] in patients with celiac disease who are taking a gluten-containing diet are:
- Increased density of intraepithelial lymphocytes (> 25/100 epithelial cells)
- Crypt hyperplasia, with a decreased villi/crypt ratio
- Blunted or atrophic villi
- Mononuclear cell infiltration into the lamina propria
- Epithelial changes, including structural abnormalities in epithelial cells
It is highly recommended that the pathologist’s report should include changes in a structured format, including the above-mentioned histological changes, intraepithelial lymphocyte count, and interpretation in terms of the modified Marsh classification. Pathologists should also comment on the quality of biopsies. If biopsies are of poor quality, with small unorientated fragments, the pathologist should abstain from making a diagnosis. In these cases, endoscopy with multiple biopsies should be repeated before gluten restriction.
A series of well-designed studies by Marsh [20] made it possible to interpret the wide range of mucosal damage induced by gluten, with the celiac histological modifications being categorized as ranging from normal mucosa to subtotal/total villous atrophy in patients with celiac disease. The modified Marsh classification is widely used in clinical practice [73,76]. Another simpler classification has been proposed in order to minimize intraobserver variability [77].
Table 5 Modified Marsh classification of gluten-induced small-intestinal damage [73,76]
|
Stage 0
|
Pre-infiltrative mucosa; up to 30% of patients with dermatitis herpetiformis (DH) or gluten ataxia have small-intestinal biopsy specimens that appear normal
|
|
Stage 1
|
Increase in the number of intraepithelial lymphocytes (IELs) to more than 25 per 100 enterocytes with a normal crypt/villi ratio
|
|
Stage 2
|
Crypt hyperplasia. In addition to the increased IELs, there is an increase in crypt depth without a reduction in villus height
|
|
Stage 3
|
Villous effacement. This is the classic celiac disease lesion. It is found in 40% of DH patients. Despite marked mucosal changes, many individuals are asymptomatic and are therefore classified as having subclinical or silent celiac disease. This lesion is characteristic, but not pathognomonic, of celiac disease and can also be seen with severe giardiasis, infantile food allergy, or post-enteritis syndrome, graft-versus-host disease, chronic ischemia of the small intestine, tropical sprue, immunoglobulin deficiencies, and other immune deficiencies and allograft rejection
|
5.5 Serum antibody tests
Two main groups of serological markers for untreated celiac disease have been shown to be highly sensitive and specific [78,79]:
- Autoantibodies targeting the auto-antigen
— Antiendomysial (EMA) and anti-tissue transglutaminase (anti-tTG) antibodies
- Antibodies targeting the offending agent (gliadin)
— Antibodies against synthetic deamidated gliadin peptides (anti-DGPs)
All of these antibodies are based on immunoglobulin A (IgA) or immunoglobulin G (IgG). Specifically, IgG-based tests are useful for detecting celiac disease in selected IgA-deficient patients.
- Patients who have a low antibody titer and normal small-intestinal mucosa may have a false-positive test. The serological test should be repeated after 6 months while the patient is on a gluten-containing diet. If the test becomes negative, this may be regarded as transient seropositivity. There are few data on the long-term follow-up in such patients.
- If the serology remains positive, these patients may have potential celiac disease and they should be followed up and the original biopsy reviewed. The majority of potential celiac disease patients later develop celiac disease.
- In addition, 30% of patients with dermatitis herpetiformis who have positive serology findings have normal histology. However, a GFD is recommended even with negative histology, as it restores positive serology and skin lesions.
- Asymptomatic individuals with titers just at the cut-off level (borderline) should be retested after 3–6 months on a normal diet (with no gluten restriction or reduction). Symptomatic individuals with borderline serology should undergo biopsy anyway, possibly after an increase in the amount of gluten in their diet.
- Antibody tests against native gliadin (AGA-IgA or AGA-IgG) are neither sensitive nor specific for celiac disease and should not be used [62].
- Tests for measuring celiac-specific antibodies in stool, urine, and saliva have a lower performance in comparison with blood-based tests and should not be used in clinical practice.
5.5.1 IgA EMA
IgA endomysial antibodies bind to endomysium, the connective tissue located around smooth muscle, producing a characteristic staining pattern that can be visualized with indirect immunofluorescence [80]. Tests should be titrated, if possible; even low titers of serum IgA endomysial antibodies are specific for celiac disease. The test is expensive, observer-dependent, and labor-intensive, requiring expert input for correct interpretation. The target antigen has been identified as tissue transglutaminase-2. IgA endomysial antibody testing is moderately sensitive (around 80%) and has the highest specificity of all celiac disease–specific tests (with close to 100% specificity if performed by experienced operators) for untreated (active) celiac disease [62,78].
5.5.2 IgA anti-tTG
Anti-tTG antibodies are highly sensitive and specific for the diagnosis of celiac disease [81]. Enzyme-linked immunosorbent assay (ELISA) tests for IgA anti-tTG antibodies are now widely used in clinical practice. They are widely available and are easier to perform, less observer-dependent, and less expensive than the immunofluorescence assay required for detecting IgA endomysial antibodies [78,79]. Interestingly, tTG targets the same autoantigen (TG2) as EMA. The diagnostic accuracy of IgA anti-tTG assays has been further improved with the use of human tTG instead of the nonhuman tTG preparations (with poorer diagnostic accuracy) used in earlier immunoassay kits. Although anti-tTG antibodies are used throughout the world, there are still substantial differences in the performance of different commercial kits [79].
5.5.3 IgA and IgG anti-DGP antibodies
An ELISA based on the detection of synthetically developed deamidated gliadin peptides (DGPs) was introduced a few years ago, and clinical research has shown that this assay has a very high level of diagnostic accuracy in high-risk and low-risk populations. This has been confirmed in recent studies [82].
IgG-class anti-DGPs perform better than IgA-based DGP tests for diagnosing celiac disease [82–84].
5.5.4 Choosing the most appropriate serologic test in different clinical scenarios
Celiac disease–specific serological tests are important for the following two purposes [78,85]:
1. To confirm gluten dependence in patients with enteropathy (diagnosis)
- IgA-EMA, IgA anti-tTG, and IgG and IgA anti-DGP perform similarly, offering the most valuable surrogates for gluten dependence.
- IgG-based tests (anti-DGP, EMA and tTG) appear to be very helpful in IgA-deficient patients who are negative for the respective IgA-based tests.
2. To select patients for duodenal biopsy
To reduce the need for duodenal biopsies and on the basis of the different accuracy of serologic tests, a series of serological algorithms is used to select patients for biopsy in different clinical scenarios.
Choice of serological test for screening of the general population:
- Anti-tTG IgA, combined with total IgA and an IgG-based test (in those who are IgA-deficient) is the most widely used first-line algorithm for screening, due to its high sensitivity and very good specificity.
- The positive predictive value in low-risk populations depends on the titer level [86,87]. Asymptomatic individuals with low titers (e.g., less than three times the cut-off values) should be retested after 3–6 months on a diet with high gluten content before endoscopy and biopsies are considered.
- A serologic algorithm, with serial use of more specific screening assays (e.g., EMA), has therefore been widely used to improve the diagnostic accuracy in the general population [86,88].
- A combination of tests may improve case-finding. If tests are simultaneously positive, a diagnosis of celiac disease is extremely likely [85,89,90].
The EMA test requires expert observers. ELISA/radioimmunoassay-based tests for detecting IgA anti-tTG or IgG anti-DGP should therefore be recommended in settings with low expertise.
5.6 HLA-DQ2/-DQ8 genotyping
HLA-DQ2/-DQ8 genotyping may be important in diagnosing seronegative disease and to exclude celiac disease prior to embarking on a formal gluten challenge.
5.7 Presumptive gluten-free diet
In some geographic areas, such as intertropical Africa, where diagnostic facilities are lacking or unaffordable, a presumptive gluten-free diet may constitute an indirect diagnostic tool if followed by dramatic clinical improvement [29]. As such, it is easy to introduce in many areas of Africa as a low-cost, risk-free treatment. The main difficulty is convincing parents to introduce a diet for a malnourished child, and especially the lifelong duration of the diet (although this difficulty is not specific to the tropics).
Although this strategy is relevant in isolated areas, where it may be the only way to confirm and treat celiac disease, we strongly recommend against it in all settings where serology is available.
In all individuals who start a gluten-free diet (for any reason — e.g., wheat allergy, lifestyle, IBS), celiac disease should be mandatorily excluded by a negative test for celiac-specific antibodies before the diet is initiated.
5.8 Differential diagnosis
There is a wide range of clinical manifestations of celiac disease, and they may mimic the symptom complexes of many other gastrointestinal diseases. In addition, histological lesions suggestive of celiac disease are also present in many other conditions, such as:
- Tropical sprue
- Combined immunodeficiency states
- Giardiasis
- Medication-induced enteropathy (olmesartan, mycophenolate)
- HIV enteropathy
- Radiation damage
- Recent chemotherapy
- Graft-versus-host disease
- Chronic ischemia
- Cholera
- Crohn’s disease
- Helicobacter pylori infection
- Eosinophilic gastroenteritis
- Enteropathy caused by food allergy (in young children)
- Autoimmune enteropathy
- Severe malnutrition
5.9 Why should we detect celiac disease?
For symptomatic celiac disease patients, introducing a GFD can lead to significant improvement in symptoms, abnormal biochemical measures, and quality of life [91]. Lifelong treatment also reduces the risk of malignant and nonmalignant complications. Concerns remain about the long-term consequences in patients with asymptomatic celiac disease, and whether maintaining a lifelong GFD is necessary for all patients. Several studies suggest that patients who have the condition detected during screening, most of whom can be regarded as asymptomatic, can improve their quality of life in the longer term with a GFD [92,93].
Patients with (long-term untreated) celiac disease have an elevated risk for benign and malignant complications, and mortality [64,94,95].
- Cancer — highest risk in the initial years after diagnosis, decreases to (near) normal risk by the fifth year [96], overall risk increment 1.35
- Malignant lymphomas
- Small-bowel adenocarcinoma
- Oropharyngeal tumors
- Unexplained infertility (12%)
- Impaired bone health and growth (osteoporosis 30–40%)
- Bone fractures — increased risk 35% for classically symptomatic celiac disease patients [97,98]
- The mortality risk is elevated in adult celiac patients, due to an increased risk for fatal malignancy (hazard ratio, 1.31; 95% confidence intervals, 1.13 to 1.51 in one study) [64]
- Adverse pregnancy outcome [99]
6. Management of celiac disease
The only treatment for celiac disease, at present, is a strictly gluten-free diet for life [20,66,100,101]. No foods or medications containing gluten from wheat, rye, or barley or their derivatives can be taken, as even small quantities of gluten may be harmful.
- Removal of gluten (with a reduction to below 20 mg gluten intake per day) from the diet of celiac disease patients will result in symptomatic, serologic, and histological remission in most patients [84,93].
- Approximately 70% of patients report an improvement in symptoms within 2 weeks of starting the gluten-free diet [93].
- Growth and development in children return to normal with adherence to a gluten-free diet. Many complications of the disease are preventable with a gluten-free diet [102,103].
- With strict dietary adherence, the titer of celiac disease–specific antibodies normalizes.
- Although the villous changes start to improve within months after the start of a gluten-free diet, complete histological resolution may take years and may not be achieved in every patient [104,105]. There is evidence that a lack of histological resolution may be dependent on persistent consumption of gluten [64].
The safe limit of gluten intake varies from patient to patient and has been considered to be 10–100 mg/day, although a subsequent study indicated that the upper limit should be no more than 50 mg/day [106].
- Pure, uncontaminated oats are not toxic in over 95% of patients with celiac disease, and have been used as part of GFDs in some countries (such as Finland) in adults and children for more than 15 years without an increased risk for enteropathy. There is a very small subgroup (< 5%) in whom oats are not safe [107,108]. There is reluctance in some countries to advise liberal use of oats, because of difficulties in guaranteeing that commercially available oats will be free of contamination with other grains. An oat-free diet, at least during the first months of treatment, can therefore be recommended.
- Rice and corn (maize) can be included in a gluten-free diet.
Although most patients have a rapid clinical response to a gluten-free diet, the rate of response varies. Patients who are extremely ill may require hospital admission, repletion of fluids and electrolytes, intravenous nutrition, iron, vitamins, and occasionally, steroids [109].
Patients should be encouraged to eat natural high-iron and high-folate foods, especially if a deficiency in these minerals is documented. Patients should consult a dietitian who is knowledgeable about gluten-free diets, especially during the first year after diagnosis. Not all dietitians are familiar with the complexity of a gluten-free diet, and local or national support groups may provide most of the information required.
Patient support and education at the time of diagnosis are very important and should be an integral part of the management [110].
Adolescents with celiac disease should undergo a formal transition process, with education and a structured transfer to adult care [111].
6.1 Recommendations after diagnosis
When following up celiac disease, it is important to consider the implications of the following concepts at the moment of evaluating the GFD:
- Serological tests cannot detect minimal gluten intake (traces), so an interdisciplinary approach is needed for detailed evaluation of the clinical situation and the GFD. This evaluation should be done by expert physicians and nutritionists [93,112].
- The need for repeated duodenal biopsies to evaluate healing and assess adherence to a GFD is a controversial matter among experts. Although this approach is frequently used in practice, it is not clear whether it is necessary for patients who respond to the GFD clinically and have decreasing or negative autoantibody levels. Among the experts who recommend repeated biopsy, the timing for when samples should be obtained is not well defined. Complete healing of the intestinal mucosa is also often slow or incomplete, especially among adults. Intestinal biopsies should be considered mandatory in patients with persistent symptoms despite evidence of a strict GFD.
- Continuing consumption of high amounts of gluten as well as gluten traces (intentional or unnoticed) is the most likely cause in patients who do not respond to the treatment [108,113,114].
The following is a summary of recommendations for follow-up after diagnosis and tools for monitoring adherence to a gluten-free diet [9] during the first year after diagnosis:
Clinical evaluation: check symptoms and do laboratory tests (e.g., full blood count, iron status, vitamin B12, celiac serology, calcium, vitamin D) and celiac disease serology tests (best predictors so far: quantitative determination of anti-tTG IgA and anti-DGP IgA) [84,104,115].
- Children and adolescents should be monitored annually (anthropometry, pubertal development, celiac-specific serology) by a pediatrician or pediatric gastroenterologist until they have reached their final height.
- A bone mineral density scan can be performed to provide a baseline measure of bone mass [116].
- Vaccination against pneumococci, Haemophilus influenzae, meningococci [117].
- Counseling by a nutritionist: assessment of nutritional status and adherence to a gluten-free diet on the basis of an interview, a food diary, and frequency of consumption (coinciding with the clinical visit).
- The patient should be helped to join support groups with acknowledged experience in celiac disease — levels of adherence to gluten-free diet are associated with membership of advocacy groups and regular dietetic follow-up [118,119].
- If necessary and/or requested, a psychological consultation should be offered [120].
6.2 Gluten-free diet
6.2.1 Not allowed in a gluten-free diet
Grains, starches, and flours not permitted in a gluten-free diet [101,121]:
- Barley
- Bran
- Bulgur
- Couscous
- Durum flour
- Einkorn (Triticum monococcum)*
- Emmer (Triticum dicoccum)*
- Farro*
- Gluten, gluten flour
- Graham flour
- Kamut™ (Khorasan wheat)*
- Malt, malt extract, malt flavoring, malt syrup
- Oats, oat bran, oat syrup — not labeled as gluten-free
- Rye
- Semolina (durum wheat)*
- Spelt (dinkel wheat, Triticum spelta)
- Triticale
- Wheat germ, wheat starch, wheat bran
- Any item with wheat, barley, or rye in its name
* Varieties of wheat
Pure oats free of contamination are available in some countries, and these are allowed in certain quantities. Although many studies have indicated that a moderate amount of oats can safety be eaten by patients with celiac disease, there are concerns regarding the contamination of oats with wheat and barley during processing.
6.2.2 Allowed in a gluten-free diet
Gluten-free grains, flours, and starches that are allowed in a gluten-free diet:
- Amaranth
- Arrowroot
- Bean flours
- Buckwheat
- Corn
- Garbanzo beans
- Seeds
- Millet
- Montina flour (Indian rice grass)
- Nut flour and nut meals
- Oats (uncontaminated, labeled as gluten-free)
- Potato flour, potato starch
- Quinoa
- Rice, all forms (brown, white, sweet, wild, jasmine, basmati, glutinous rice, polished rice, rice bran)
- Sorghum flour
- Soy flour
- Tapioca
- Teff flour
Although these gluten-free grains, flours, and starches are allowed in a GFD, there are concerns regarding cross-contamination with wheat and barley. Starches and flours should therefore be analyzed for gluten content before permitted use in diets for celiac disease patients. Some countries periodically publish updated lists of gluten-free products on the Internet. Foods on the list have been analyzed for gluten content [122,123].
6.2.3 Other foods for a basic gluten-free diet
- Dairy
- All fresh meats and gluten-free preserved meat
- Seafood
- Eggs
- Legumes: lentils, chickpeas (garbanzo beans), peas, beans, nuts, seeds —packaged, not loose
- Fruits (fresh, frozen, canned) and plain fruit juices
- Vegetables (fresh, frozen, canned) and plain vegetable juices
- Liquid vegetable oils
6.2.4 Miscellaneous items allowed in a gluten-free diet
- Sweets: honey, corn syrup, sugar (brown and white)
- Snack foods: plain popcorn, nuts, plain pickles, olives, gluten-free potato chips (potato crisps)
- Condiments: natural herbs, pure black pepper, vinegars (apple, grape, or wine)
Cooking and food preparation: patients should be instructed not to contaminate gluten-free food — e.g., using separate cooking utensils, cooking surfaces, and toasters.
The majority of industrially produced foods contain nonallowable ingredients — attention to labeling is important, and available lists should be checked for allowable foodstuffs. It is very important to access a support group.
A gluten-free diet is low in fiber. Patients should be advised to eat a high-fiber diet supplemented with whole-grain rice, maize, potatoes, and ample vegetables. Any dietary deficiencies such as iron, folic acid, calcium and (very rarely) vitamin B12 should be corrected.
6.3 Prevention — screening
Currently, there are no definite measures for the primary prevention of celiac disease based on the timing of gluten introduction for infants or the duration of breastfeeding. Randomized controlled trials have failed to induce tolerance by feeding small amounts of gluten [48] or delaying gluten introduction until after 12 months of age [49]. Whether the amount of gluten is important is still a matter of controversy [51,124,125]. Early detection and treatment are the most valid methods for secondary prevention of celiac disease [126].
In the debate on how to improve the detection of gluten-related disorders, some recommend switching from high risk–based case-finding to an active policy of serological celiac disease testing in the general population [68,127].
6.4 New management options
As the steps involved in the immunopathogenesis of celiac disease have been gradually unraveled, many immunopathogenetic targets are now being explored for alternative/adjunct treatments, such as intraluminal digestion of gluten, improvement in barrier functions, and immunomodulators. Many of the new options have reached the stage of clinical trials, but none of them is at present available for clinical use.
6.5 Monitoring
6.5.1 Laboratory assessment
Celiac serology can be useful, but falling concentrations of celiac-specific antibodies indicate gluten reduction and have limited ability to define complete adherence. Once the antibodies have normalized, a subsequent increase in level is considered a good indicator of gluten ingestion. The ultimate measure of adherence is the demonstration of intestinal healing, but this may not occur even in patients with strict gluten avoidance.
Assessment interval: every 3–6 months until abnormal baseline tests are corrected or until clinical stabilization; then every 1–2 years.
- Studies suggest that periodic testing for IgA anti-tTG or IgA anti-DGP is the preferred method for monitoring compliance.
- Although these tests do not identify minor dietary indiscretions — normal titers are insensitive for ongoing gluten exposure or enteropathy — a continued reduction in serum concentrations helps assess compliance with the diet.
- Persistent borderline, increased, or increasing titers indicate significant gluten exposure [64].
6.5.2 Nutritionist consultation
Interval: ideally every 3–6 months until clinical normalization; then every 1–2 years. This is particularly important in women of child-bearing age and during pregnancy. An expert dietitian should be consulted in order to:
- Assess the patient’s current nutritional status.
- Identify macronutrient and/or micronutrient intake and detect deficiencies and/or excesses.
— It is important that patients with celiac disease consume a well-balanced diet including vitamins, calcium, and fibers, with specialist consultation.
- Analyze eating habits and potential factors affecting access to the diet.
- Provide information and initiate the gluten-free diet.
- Provide dietary education.
- Monitor and evaluate dietary compliance and reinforce alimentary counseling.
Patients who are unable to adhere to the diet may require support with psychological counseling.
6.5.3 Persistence of symptoms
Persistence of symptoms is frequently caused by continued ingestion of gluten. A common difficulty with the gluten-free diet is cross-contamination and the presence of unexpected gluten in processed food items and/or medicines. Gluten is used widely in processed food products and thus may be a hidden ingredient, so it is prudent for patients to routinely check the ingredient list before purchasing any product. If the response to gluten-free diet is suboptimal, intentional or inadvertent ingestion of gluten should be considered. In these cases, persistence of positive serology may identify major and continued lapses in dietary adherence.
List of causes of persistence of symptoms in patients with celiac disease:
- Overlapping irritable bowel syndrome or inadvertent gluten ingestion (most common causes)
- Wrong diagnosis
- Primary lactose intolerance
- Food allergies other than wheat
- Pancreatic insufficiency
- Microscopic colitis
- Bacterial overgrowth
- Inflammatory bowel disease
- Ulcerative jejunitis*
- Enteropathy-associated T-cell lymphoma*
- Refractory celiac disease*
* These can be regarded as complications of long-lasting celiac disease.
6.6 Transfer of care from childhood to adulthood
Adolescents/young adults with celiac disease are at risk of “medical” dropout prior to and during the transfer to adult health care [128,129]. In general, adults with celiac disease are used to taking care of their diet and quickly learn what they can eat. However, adolescents are often still helped by parents and caregivers and may not be autonomous in dealing with their disease. There are no precise guidelines for filling the gap between the pediatric and adult services for celiac disease, and transitional healthcare is now recommended. The transitional clinic should focus on education (dietary and health issues), the need to monitor the disease, and should deal with any psychological issues that may rise [111].
6.7 Refractory celiac disease
Refractory celiac disease is defined as persistence (or recurrence) of clinical symptoms and histopathological abnormalities in association with clinical symptoms despite excellent adherence to a GFD for at least 12 months [108]. This may occur at presentation (primary), or after an initial response to a gluten-free diet (secondary) [130]. Refractory celiac disease must be considered particularly in patients with celiac disease who are diagnosed over the age of 50.
There are two subtypes of refractory celiac disease:
- Type I, with phenotypically normal intraepithelial lymphocytes
- Type II, with clonal expansion of intraepithelial lymphocytes and an aberrant phenotype lacking surface CD3, CD8, and T-cell receptors
Type II disease is considered to be a form of low-grade intraepithelial lymphoma, revealed by severe malabsorption that is not responsive to a gluten-free diet. This is the most severe form, and it is associated with a high mortality rate [131].
7. Appendix
7.1 Abbreviations
Table 6 Abbreviations used in this WGO guideline
|
AGA
|
antigliadin antibody
|
|
ATI
|
amylase/trypsin inhibitor
|
|
CD
|
celiac disease
|
|
DGP
|
deamidated gliadin peptide
|
|
DH
|
dermatitis herpetiformis
|
|
EATL
|
enteropathy-associated T-cell lymphoma
|
|
ELISA
|
enzyme-linked immunoassay
|
|
EMA
|
anti-endomysium antibody
|
|
ESPGHAN
|
European Society for Pediatric Gastroenterology, Hepatology, and Nutrition
|
|
FODMAP
|
fermentable oligo-, di-, monosaccharides and polyol
|
|
GFD
|
gluten-free diet
|
|
GRD
|
gluten-related disorder
|
|
HIV
|
human immunodeficiency virus
|
|
HLA
|
human leukocyte antigen
|
|
IBD
|
inflammatory bowel disease
|
|
IBS
|
irritable bowl syndrome
|
|
IDDM
|
insulin-dependent diabetes mellitus
|
|
IEL
|
intraepithelial lymphocyte
|
|
IgA
|
immunoglobulin A
|
|
IgE
|
immunoglobulin E
|
|
IgG
|
immunoglobulin G
|
|
MHC
|
major histocompatibility complex
|
|
NCGS
|
nonceliac gluten sensitivity
|
|
NRCD
|
nonresponsive celiac disease
|
|
TG2
|
transglutaminase 2
|
|
tTG
|
Tissue transglutaminase
|
|
WA
|
wheat allergy
|
|
WGO
|
World Gastroenterology Organisation
|
|
WHO
|
World Health Organization
|
7.2 Gold standard guidelines
- 2016:
Transition from childhood to adulthood in coeliac disease: the Prague consensus report. Ludvigsson et al. Gut 2016;65:1242–51 [111].
- 2014:
Diagnosis and management of adult coeliac disease: guidelines from the British Society of Gastroenterology. Ludvigsson et al. Gut 2014;63:1210–28 [63].
- 2013:
ACG clinical guidelines: diagnosis and management of celiac disease. Rubio-Tapia et al. Am J Gastroenterol 2013;108:656–76; quiz 677. doi: 10.1038/ajg.2013.79 [132].
- 2012:
European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of coeliac disease. Husby et al. J Pediatr Gastroenterol Nutr 2012;54:136–60 [87].
- 2005:
Guideline for the diagnosis and treatment of celiac disease in children: recommendations of the North American Society for Pediatric Gastroenterology, Hepatology and Nutrition. Hill et al. J Pediatr Gastroenterol Nutr 2005;40:1–19 [67].
References
- Stern M, Ciclitira PJ, van Eckert R, Feighery C, Janssen FW, Méndez E, et al. Analysis and clinical effects of gluten in coeliac disease. Eur J Gastroenterol Hepatol 2001;13(6):741–7.
- Jabri B, Sollid LM. Mechanisms of disease: immunopathogenesis of celiac disease. Nat Clin Pract Gastroenterol Hepatol 2006;3(9):516–25.
- Ludvigsson JF, Leffler DA, Bai JC, Biagi F, Fasano A, Green PHR, et al. The Oslo definitions for coeliac disease and related terms. Gut 2013;62(1):43–52.
- Cianferoni A. Wheat allergy: diagnosis and management. J Asthma Allergy 2016;13.
- Sapone A, Bai JC, Ciacci C, Dolinsek J, Green PHR, Hadjivassiliou M, et al. Spectrum of gluten-related disorders: consensus on new nomenclature and classification. BMC Med 2012;10:13.
- Carroccio A, Mansueto P, Iacono G, Soresi M, D’Alcamo A, Cavataio F, et al. Non-celiac wheat sensitivity diagnosed by double-blind placebo-controlled challenge: exploring a new clinical entity. Am J Gastroenterol 2012;107(12):1898–906.
- Biesiekierski JR, Newnham ED, Irving PM, Barrett JS, Haines M, Doecke JD, et al. Gluten causes gastrointestinal symptoms in subjects without celiac disease: a double-blind randomized placebo-controlled trial. Am J Gastroenterol 2011;106(3):508–14.
- Biesiekierski JR, Peters SL, Newnham ED, Rosella O, Muir JG, Gibson PR. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology 2013;145(2):320–328.e3.
- Di Sabatino A, Volta U, Salvatore C, Biancheri P, Caio G, De Giorgio R, et al. Small amounts of gluten in subjects with suspected nonceliac gluten sensitivity: a randomized, double-blind, placebo-controlled, cross-over trial. Clin Gastroenterol Hepatol 2015;13(9):1604–1612.e3.
- Shahbazkhani B, Sadeghi A, Malekzadeh R, Khatavi F, Etemadi M, Kalantri E, et al. Non-celiac gluten sensitivity has narrowed the spectrum of irritable bowel syndrome: a double-blind randomized placebo-controlled trial. Nutrients 2015;7(6):4542–54.
- Carroccio A, D’Alcamo A, Mansueto P. Nonceliac wheat sensitivity in the context of multiple food hypersensitivity: new data from confocal endomicroscopy. Gastroenterology 2015;148(3):666–7.
- Guandalini S, Polanco I. Nonceliac gluten sensitivity or wheat intolerance syndrome? J Pediatr 2015;166(4):805–11.
- Lohi S, Mustalahti K, Kaukinen K, Laurila K, Collin P, Rissanen H, et al. Increasing prevalence of coeliac disease over time. Aliment Pharmacol Ther 2007;26(9):1217–25.
- Choung RS, Ditah IC, Nadeau AM, Rubio-Tapia A, Marietta EV, Brantner TL, et al. Trends and racial/ethnic disparities in gluten-sensitive problems in the United States: findings from the National Health and Nutrition Examination Surveys from 1988 to 2012. Am J Gastroenterol 2015;110(3):455–61.
- Fasano A, Catassi C. Current approaches to diagnosis and treatment of celiac disease: an evolving spectrum. Gastroenterology 2001;120(3):636–51.
- Fasano A, Berti I, Gerarduzzi T, Not T, Colletti RB, Drago S, et al. Prevalence of celiac disease in at-risk and not-at-risk groups in the United States: a large multicenter study. Arch Intern Med 2003;163(3):286–92.
- Lionetti E, Catassi C. New clues in celiac disease epidemiology, pathogenesis, clinical manifestations, and treatment. Int Rev Immunol 2011;30(4):219–31.
- Tortora R, Zingone F, Rispo A, Bucci C, Capone P, Imperatore N, et al. Coeliac disease in the elderly in a tertiary centre. Scand J Gastroenterol 2016;51(10):1179–83.
- Smecuol E, Mauriño E, Vazquez H, Pedreira S, Niveloni S, Mazure R, et al. Gynaecological and obstetric disorders in coeliac disease: frequent clinical onset during pregnancy or the puerperium. Eur J Gastroenterol Hepatol 1996;8(1):63–89.
- Marsh MN. Gluten, major histocompatibility complex, and the small intestine. A molecular and immunobiologic approach to the spectrum of gluten sensitivity (“celiac sprue”). Gastroenterology 1992;102(1):330–54.
- Gomez JC, Selvaggio GS, Viola M, Pizarro B, la Motta G, de Barrio S, et al. Prevalence of celiac disease in Argentina: screening of an adult population in the La Plata area. Am J Gastroenterol 2001;96(9):2700–4.
- Gandolfi L, Pratesi R, Cordoba JC, Tauil PL, Gasparin M, Catassi C. Prevalence of celiac disease among blood donors in Brazil. Am J Gastroenterol 2000;95(3):689–92.
- Parada A, Araya M, Pérez-Bravo F, Méndez M, Mimbacas A, Motta P, et al. Amerindian mtDNA haplogroups and celiac disease risk HLA haplotypes in mixed-blood Latin American patients. J Pediatr Gastroenterol Nutr 2011;53(4):429–34.
- Barada K, Abu Daya H, Rostami K, Catassi C. Celiac disease in the developing world. Gastrointest Endosc Clin N Am 2012;22(4):773–96.
- Brar P, Lee AR, Lewis SK, Bhagat G, Green PHR. Celiac disease in African-Americans. Dig Dis Sci 2006;51(5):1012–5.
- Catassi C, Rätsch IM, Gandolfi L, Pratesi R, Fabiani E, El Asmar R, et al. Why is coeliac disease endemic in the people of the Sahara? Lancet 1999;354(9179):647–8.
- Sood A, Midha V, Sood N, Avasthi G, Sehgal A. Prevalence of celiac disease among school children in Punjab, North India. J Gastroenterol Hepatol 2006;21(10):1622–5.
- Aziz S, Muzaffar R, Zafar MN, Mehnaz A, Mubarak M, Abbas Z, et al. Celiac disease in children with persistent diarrhea and failure to thrive. J Coll Physicians Surg Pak 2007;17(9):554–7.
- Diallo I, Coton T. Celiac disease: a challenging disease uneasy to diagnose in Sub-Saharan Africa. J Gastroenterol Hepatol Res [Internet] 2013;2(8):753–6. Available at: http://www.ghrnet.org/index.php/joghr/article/view/459.
- Ageep AK. Celiac disease in the Red Sea state of Sudan. Trop Gastroenterol 2012;33(2):118–22.
- Mohammed IM, Karrar ZEA, El-Safi SH. Coeliac disease in Sudanese children with clinical features suggestive of the disease. East Mediterr Health J 2006;12(5):582–9.
- Wu J, Xia B, von Blomberg BME, Zhao C, Yang XW, Crusius JBA, et al. Coeliac disease: emerging in China? Gut 2010;59(3):418–9.
- Singh P, Arora S, Singh A, Strand TA, Makharia GK. Prevalence of celiac disease in Asia: a systematic review and meta-analysis. J Gastroenterol Hepatol 2016;31(6):1095–101.
- Vázquez H, de la Paz Temprano M, Sugai E, Scacchi SM, Souza C, Cisterna D, et al. Prevalence of celiac disease and celiac autoimmunity in the Toba native Amerindian community of Argentina. Can J Gastroenterol Hepatol 2015;29(8):431–4.
- Lionetti E, Gatti S, Pulvirenti A, Catassi C. Celiac disease from a global perspective. Best Pract Res Clin Gastroenterol 2015;29(3):365–79.
- Catassi C, Kryszak D, Bhatti B, Sturgeon C, Helzlsouer K, Clipp SL, et al. Natural history of celiac disease autoimmunity in a USA cohort followed since 1974. Ann Med 2010;42(7):530–8.
- Mäki M, Mustalahti K, Kokkonen J, Kulmala P, Haapalahti M, Karttunen T, et al. Prevalence of Celiac disease among children in Finland. N Engl J Med 2003;348(25):2517–24.
- Green PHR. The many faces of celiac disease: clinical presentation of celiac disease in the adult population. Gastroenterology 2005;128(4 Suppl 1):S74-78.
- Dickey W, McMillan SA, Hughes DF. Identification of coeliac disease in primary care. Scand J Gastroenterol 1998;33(5):491–3.
- Collin P, Huhtala H, Virta L, Kekkonen L, Reunala T. Diagnosis of celiac disease in clinical practice: physician’s alertness to the condition essential. J Clin Gastroenterol 2007;41(2):152–6.
- Rostami-Nejad M, Romanos J, Rostami K, Ganji A, Ehsani-Ardakani MJ, Bakhshipour AR, et al. Allele and haplotype frequencies for HLA-DQ in Iranian celiac disease patients. World J Gastroenterol 2014;20(20):6302–8.
- Gutierrez-Achury J, Zhernakova A, Pulit SL, Trynka G, Hunt KA, Romanos J, et al. Fine mapping in the MHC region accounts for 18% additional genetic risk for celiac disease. Nat Genet 2015;47(6):577–8.
- Romanos J, van Diemen CC, Nolte IM, Trynka G, Zhernakova A, Fu J, et al. Analysis of HLA and non-HLA alleles can identify individuals at high risk for celiac disease. Gastroenterology 2009;137(3):834–40, 840–3.
- Liu E, Lee HS, Aronsson CA, Hagopian WA, Koletzko S, Rewers MJ, et al. Risk of pediatric celiac disease according to HLA haplotype and country. N Engl J Med 2014;371(1):42–9.
- Polvi A, Arranz E, Fernandez-Arquero M, Collin P, Mäki M, Sanz A, et al. HLA-DQ2-negative celiac disease in Finland and Spain. Hum Immunol 1998;59(3):169–75.
- Spurkland A, Sollid LM, Polanco I, Vartdal F, Thorsby E. HLA-DR and -DQ genotypes of celiac disease patients serologically typed to be non-DR3 or non-DR5/7. Hum Immunol 1992;35(3):188–92.
- Abadie V, Sollid LM, Barreiro LB, Jabri B. Integration of genetic and immunological insights into a model of celiac disease pathogenesis. Annu Rev Immunol 2011;29:493–525.
- Vriezinga SL, Auricchio R, Bravi E, Castillejo G, Chmielewska A, Crespo Escobar P, et al. Randomized feeding intervention in infants at high risk for celiac disease. N Engl J Med 2014;371(14):1304–15.
- Lionetti E, Castellaneta S, Francavilla R, Pulvirenti A, Tonutti E, Amarri S, et al. Introduction of gluten, HLA status, and the risk of celiac disease in children. N Engl J Med 2014;371(14):1295–303.
- Aronsson CA, Lee HS, Liu E, Uusitalo U, Hummel S, Yang J, et al. Age at gluten introduction and risk of celiac disease. Pediatrics 2015;135(2):239–45.
- Andrén Aronsson C, Lee HS, Koletzko S, Uusitalo U, Yang J, Virtanen SM, et al. Effects of gluten intake on risk of celiac disease: a case-control study on a Swedish birth cohort. Clin Gastroenterol Hepatol 2016;14(3):403–409.e3.
- Szajewska H, Shamir R, Chmielewska A, Pieścik-Lech M, Auricchio R, Ivarsson A, et al. Systematic review with meta-analysis: early infant feeding and coeliac disease—update 2015. Aliment Pharmacol Ther 2015;41(11):1038–54.
- Ferguson A, Arranz E, O’Mahony S. Clinical and pathological spectrum of coeliac disease—active, silent, latent, potential. Gut 1993;34(2):150–1.
- Green PHR, Jabri B. Coeliac disease. Lancet 2003;362(9381):383–91.
- Santonicola A, Iovino P, Cappello C, Capone P, Andreozzi P, Ciacci C. From menarche to menopause: the fertile life span of celiac women. Menopause 2011;18(10):1125–30.
- Sbai W, Bourgain G, Luciano L, Brardjanian S, Thefenne L, Al Shukry A, et al. Celiac crisis in a multi-trauma adult patient. Clin Res Hepatol Gastroenterol 2016;40(3):e31–2.
- Korpimäki S, Kaukinen K, Collin P, Haapala A-M, Holm P, Laurila K, et al. Gluten-sensitive hypertransaminasemia in celiac disease: an infrequent and often subclinical finding. Am J Gastroenterol 2011;106(9):1689–96.
- Nachman F, Mauriño E, Vázquez H, Sfoggia C, Gonzalez A, Gonzalez V, et al. Quality of life in celiac disease patients: prospective analysis on the importance of clinical severity at diagnosis and the impact of treatment. Dig Liver Dis 2009;41(1):15–25.
- Lindfors K, Koskinen O, Kaukinen K. An update on the diagnostics of celiac disease. Int Rev Immunol 2011;30(4):185–96.
- Kinos S, Kurppa K, Ukkola A, Collin P, Lähdeaho ML, Huhtala H, et al. Burden of illness in screen-detected children with celiac disease and their families. J Pediatr Gastroenterol Nutr 2012;55(4):412–6.
- Kurppa K, Paavola A, Collin P, Sievänen H, Laurila K, Huhtala H, et al. Benefits of a gluten-free diet for asymptomatic patients with serologic markers of celiac disease. Gastroenterology 2014;147(3):610–617.e1.
- Giersiepen K, Lelgemann M, Stuhldreher N, Ronfani L, Husby S, Koletzko S, et al. Accuracy of diagnostic antibody tests for coeliac disease in children: summary of an evidence report. J Pediatr Gastroenterol Nutr 2012;54(2):229–41.
- Ludvigsson JF, Bai JC, Biagi F, Card TR, Ciacci C, Ciclitira PJ, et al. Diagnosis and management of adult coeliac disease: guidelines from the British Society of Gastroenterology. Gut 2014;63(8):1210–28.
- Kelly CP, Bai JC, Liu E, Leffler DA. Advances in diagnosis and management of celiac disease. Gastroenterology 2015;148(6):1175–86.
- Catassi C, Fasano A. Celiac disease diagnosis: simple rules are better than complicated algorithms. Am J Med 2010;123(8):691–3.
- Ciclitira PJ, King AL, Fraser JS. AGA technical review on Celiac Sprue. American Gastroenterological Association. Gastroenterology 2001;120(6):1526–40.
- Hill ID, Dirks MH, Liptak GS, Colletti RB, Fasano A, Guandalini S, et al. Guideline for the diagnosis and treatment of celiac disease in children: recommendations of the North American Society for Pediatric Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr 2005;40(1):1–19.
- Ludvigsson JF, Card TR, Kaukinen K, Bai J, Zingone F, Sanders DS, et al. Screening for celiac disease in the general population and in high-risk groups. United Eur Gastroenterol J 2015;3(2):106–20.
- Singh P, Arora S, Lal S, Strand TA, Makharia GK. Risk of celiac disease in the first- and second-degree relatives of patients with celiac disease: a systematic review and meta-analysis. Am J Gastroenterol 2015;110(11):1539–48.
- Oxentenko AS, Grisolano SW, Murray JA, Burgart LJ, Dierkhising RA, Alexander JA. The insensitivity of endoscopic markers in celiac disease. Am J Gastroenterol 2002;97(4):933–8.
- Niveloni S, Fiorini A, Dezi R, Pedreira S, Smecuol E, Vazquez H, et al. Usefulness of videoduodenoscopy and vital dye staining as indicators of mucosal atrophy of celiac disease: assessment of interobserver agreement. Gastrointest Endosc 1998;47(3):223–9.
- Marsh MN, W Johnson M, Rostami K. Mucosal histopathology in celiac disease: a rebuttal of Oberhuber’s sub-division of Marsh III. Gastroenterol Hepatol Bed Bench 2015;8(2):99–109.
- Oberhuber G, Granditsch G, Vogelsang H. The histopathology of coeliac disease: time for a standardized report scheme for pathologists. Eur J Gastroenterol Hepatol 1999;11(10):1185–94.
- Mooney PD, Kurien M, Evans KE, Rosario E, Cross SS, Vergani P, et al. Clinical and immunologic features of ultra-short celiac disease. Gastroenterology 2016;150(5):1125–34.
- Leffler D, Schuppan D, Pallav K, Najarian R, Goldsmith JD, Hansen J, et al. Kinetics of the histological, serological and symptomatic responses to gluten challenge in adults with coeliac disease. Gut 2013;62(7):996–1004.
- Rostami K, Kerckhaert J, Tiemessen R, von Blomberg BM, Meijer JW, Mulder CJ. Sensitivity of antiendomysium and antigliadin antibodies in untreated celiac disease: disappointing in clinical practice. Am J Gastroenterol 1999;94(4):888–94.
- Corazza GR, Villanacci V. Coeliac disease. J Clin Pathol 2005;58(6):573–4.
- Rostom A, Dubé C, Cranney A, Saloojee N, Sy R, Garritty C, et al. The diagnostic accuracy of serologic tests for celiac disease: a systematic review. Gastroenterology 2005;128(4 Suppl 1):S38-46.
- Leffler DA, Schuppan D. Update on serologic testing in celiac disease. Am J Gastroenterol 2010;105(12):2520–4.
- Chorzelski TP, Beutner EH, Sulej J, Tchorzewska H, Jablonska S, Kumar V, et al. IgA anti-endomysium antibody. A new immunological marker of dermatitis herpetiformis and coeliac disease. Br J Dermatol 1984;111(4):395–402.
- Dieterich W, Ehnis T, Bauer M, Donner P, Volta U, Riecken EO, et al. Identification of tissue transglutaminase as the autoantigen of celiac disease. Nat Med 1997;3(7):797–801.
- Sugai E, Moreno ML, Hwang HJ, Cabanne A, Crivelli A, Nachman F, et al. Celiac disease serology in patients with different pretest probabilities: is biopsy avoidable? World J Gastroenterol 2010;16(25):3144–52.
- Bufler P, Heilig G, Ossiander G, Freudenberg F, Grote V, Koletzko S. Diagnostic performance of three serologic tests in childhood celiac disease. Z Gastroenterol 2015;53(2):108–14.
- Nachman F, Sugai E, Vázquez H, González A, Andrenacci P, Niveloni S, et al. Serological tests for celiac disease as indicators of long-term compliance with the gluten-free diet. Eur J Gastroenterol Hepatol 2011;23(6):473–80.
- Weile B, Hansen BF, Hägerstrand I, Hansen JP, Krasilnikoff PA. Interobserver variation in diagnosing coeliac disease. A joint study by Danish and Swedish pathologists. APMIS 2000;108(5):380–4.
- Webb C, Norström F, Myléus A, Ivarsson A, Halvarsson B, Högberg L, et al. Celiac disease can be predicted by high levels of anti-tissue transglutaminase antibodies in population-based screening. J Pediatr Gastroenterol Nutr 2015;60(6):787–91.
- Husby S, Koletzko S, Korponay-Szabó IR, Mearin ML, Phillips A, Shamir R, et al. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of coeliac disease. J Pediatr Gastroenterol Nutr 2012;54(1):136–60.
- Sandström O, Rosén A, Lagerqvist C, Carlsson A, Hernell O, Högberg L, et al. Transglutaminase IgA antibodies in a celiac disease mass screening and the role of HLA-DQ genotyping and endomysial antibodies in sequential testing. J Pediatr Gastroenterol Nutr 2013;57(4):472–6.
- Sugai E, Hwang HJ, Vázquez H, Moreno ML, Costa F, Longarini G, et al. Should ESPGHAN guidelines for serologic diagnosis of celiac disease be used in adults? A prospective analysis in an adult patient cohort with high pretest probability. Am J Gastroenterol 2015;110(10):1504–5.
- Oyaert M, Vermeersch P, De Hertogh G, Hiele M, Vandeputte N, Hoffman I, et al. Combining antibody tests and taking into account antibody levels improves serologic diagnosis of celiac disease. Clin Chem Lab Med 2015;53(10):1537–46.
- Newnham ED, Shepherd SJ, Strauss BJ, Hosking P, Gibson PR. Adherence to the gluten-free diet can achieve the therapeutic goals in almost all patients with coeliac disease: a 5-year longitudinal study from diagnosis. J Gastroenterol Hepatol 2016;31(2):342–9.
- Kurppa K, Collin P, Mäki M, Kaukinen K. Celiac disease and health-related quality of life. Expert Rev Gastroenterol Hepatol 2011;5(1):83–90.
- Nachman F, del Campo MP, González A, Corzo L, Vázquez H, Sfoggia C, et al. Long-term deterioration of quality of life in adult patients with celiac disease is associated with treatment noncompliance. Dig Liver Dis 2010;42(10):685–91.
- Cranney A, Rostom A, Sy R, Dubé C, Saloogee N, Garritty C, et al. Consequences of testing for celiac disease. Gastroenterology 2005;128(4 Suppl 1):S109–20.
- Brousse N, Meijer JWR. Malignant complications of coeliac disease. Best Pract Res Clin Gastroenterol 2005;19(3):401–12.
- Anderson LA, Gadalla S, Morton LM, Landgren O, Pfeiffer R, Warren JL, et al. Population-based study of autoimmune conditions and the risk of specific lymphoid malignancies. Int J Cancer 2009;125(2):398–405.
- Corazza GR, Di Stefano M, Mauriño E, Bai JC. Bones in coeliac disease: diagnosis and treatment. Best Pract Res Clin Gastroenterol 2005;19(3):453–65.
- Olmos M, Antelo M, Vazquez H, Smecuol E, Mauriño E, Bai JC. Systematic review and meta-analysis of observational studies on the prevalence of fractures in coeliac disease. Dig Liver Dis 2008;40(1):46–53.
- Saccone G, Berghella V, Sarno L, Maruotti GM, Cetin I, Greco L, et al. Celiac disease and obstetric complications: a systematic review and metaanalysis. Am J Obstet Gynecol 2016;214(2):225–34.
- Green PHR, Cellier C. Celiac disease. N Engl J Med 2007;357(17):1731–43.
- Ciacci C, Ciclitira P, Hadjivassiliou M, Kaukinen K, Ludvigsson JF, McGough N, et al. The gluten-free diet and its current application in coeliac disease and dermatitis herpetiformis. United Eur Gastroenterol J 2015;3(2):121–35.
- Collin P. Should adults be screened for celiac disease? What are the benefits and harms of screening? Gastroenterology 2005;128(4 Suppl 1):S104–8.
- Hoffenberg EJ. Should all children be screened for celiac disease? Gastroenterology 2005;128(4 Suppl 1):S98–103.
- Sugai E, Nachman F, Váquez H, González A, Andrenacci P, Czech A, et al. Dynamics of celiac disease-specific serology after initiation of a gluten-free diet and use in the assessment of compliance with treatment. Dig Liver Dis 2010;42(5):352–8.
- Moreno M de L, Cebolla Á, Muñoz-Suano A, Carrillo-Carrion C, Comino I, Pizarro Á, et al. Detection of gluten immunogenic peptides in the urine of patients with coeliac disease reveals transgressions in the gluten-free diet and incomplete mucosal healing. Gut 2015 Nov 25. doi: 10.1136/gutjnl-2015-310148 [Epub ahead of print].
- Forbes GM. If only my celiac patients and I knew that…. Clin Gastroenterol Hepatol 2015;13(3):614–5.
- Pulido OM, Gillespie Z, Zarkadas M, Dubois S, Vavasour E, Rashid M, et al. Introduction of oats in the diet of individuals with celiac disease: a systematic review. Adv Food Nutr Res 2009;57:235–85.
- Akobeng AK, Thomas AG. Systematic review: tolerable amount of gluten for people with coeliac disease. Aliment Pharmacol Ther 2008;27(11):1044–52.
- Ciacci C, Maiuri L, Russo I, Tortora R, Bucci C, Cappello C, et al. Efficacy of budesonide therapy in the early phase of treatment of adult coeliac disease patients with malabsorption: an in vivo/in vitro pilot study. Clin Exp Pharmacol Physiol 2009;36(12):1170–6.
- Ludvigsson JF, Card T, Ciclitira PJ, Swift GL, Nasr I, Sanders DS, et al. Support for patients with celiac disease: a literature review. United Eur Gastroenterol J 2015;3(2):146–59.
- Ludvigsson JF, Agreus L, Ciacci C, Crowe SE, Geller MG, Green PHR, et al. Transition from childhood to adulthood in coeliac disease: the Prague consensus report. Gut 2016;65(8):1242–51.
- Haines ML, Anderson RP, Gibson PR. Systematic review: the evidence base for long-term management of coeliac disease. Aliment Pharmacol Ther 2008;28(9):1042–66.
- Leffler DA, Edwards George JB, Dennis M, Cook EF, Schuppan D, Kelly CP. A prospective comparative study of five measures of gluten-free diet adherence in adults with coeliac disease. Aliment Pharmacol Ther 2007;26(9):1227–35.
- Hollon JR, Cureton PA, Martin ML, Puppa ELL, Fasano A. Trace gluten contamination may play a role in mucosal and clinical recovery in a subgroup of diet-adherent non-responsive celiac disease patients. BMC Gastroenterol 2013;13:40.
- Ciacci C, Cavallaro R, della Valle N, d’Argenio G. The use of serum tTG-ab assay in patients on gluten-free diet as a measure of dietetic compliance. Gastroenterology 2002;122(2):588.
- García-Manzanares A, Tenias JM, Lucendo AJ. Bone mineral density directly correlates with duodenal Marsh stage in newly diagnosed adult celiac patients. Scand J Gastroenterol 2012;47(8–9):927–36.
- Zingone F, Abdul Sultan A, Crooks CJ, Tata LJ, Ciacci C, West J. The risk of community-acquired pneumonia among 9803 patients with coeliac disease compared to the general population: a cohort study. Aliment Pharmacol Ther 2016;44(1):57–67.
- Hall NJ, Rubin G, Charnock A. Systematic review: adherence to a gluten-free diet in adult patients with coeliac disease. Aliment Pharmacol Ther 2009;30(4):315–30.
- Leffler DA, Edwards-George J, Dennis M, Schuppan D, Cook F, Franko DL, et al. Factors that influence adherence to a gluten-free diet in adults with celiac disease. Dig Dis Sci 2008;53(6):1573–81.
- Ciacci C, Iavarone A, Siniscalchi M, Romano R, De Rosa A. Psychological dimensions of celiac disease: toward an integrated approach. Dig Dis Sci 2002;47(9):2082–7.
- Niewinski MM. Advances in celiac disease and gluten-free diet. J Am Diet Assoc 2008;108(4):661–72.
- Pinto-Sanchez MI, Verdu EF, Gordillo MC, Bai JC, Birch S, Moayyedi P, et al. Tax-deductible provisions for gluten-free diet in Canada compared with systems for gluten-free diet coverage available in various countries. Can J Gastroenterol Hepatol 2015;29(2):104–10.
- Republic of Argentina. National Ministry of Health. [Clinical practice guidelines on diagnosis and treatment of celiac disease. Detection and National Celiac Disease Control. Official Gazette No. 32148]. May 2011. [Internet]. Available at: http://www.msal.gob.ar.
- Chmielewska A, Pieścik-Lech M, Szajewska H, Shamir R. Primary prevention of celiac disease: environmental factors with a focus on early nutrition. Ann Nutr Metab 2015;67 Suppl 2:43–50.
- Szajewska H, Shamir R, Mearin L, Ribes-Koninckx C, Catassi C, Domellöf M, et al. Gluten introduction and the risk of coeliac disease: a position paper by the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition. J Pediatr Gastroenterol Nutr 2016;62(3):507–13.
- Mearin ML. The prevention of coeliac disease. Best Pract Res Clin Gastroenterol 2015;29(3):493–501.
- Catassi C, Fasano A. Coeliac disease. The debate on coeliac disease screening—are we there yet? Nat Rev Gastroenterol Hepatol 2014;11(8):457–8.
- O’Leary C, Wieneke P, Healy M, Cronin C, O’Regan P, Shanahan F. Celiac disease and the transition from childhood to adulthood: a 28-year follow-up. Am J Gastroenterol 2004;99(12):2437–41.
- Mozer-Glassberg Y, Zevit N, Rosenbach Y, Hartman C, Morgenstern S, Shamir R. Follow-up of children with celiac disease—lost in translation? Digestion 2011;83(4):283–7.
- Cellier C, Delabesse E, Helmer C, Patey N, Matuchansky C, Jabri B, et al. Refractory sprue, coeliac disease, and enteropathy-associated T-cell lymphoma. French Coeliac Disease Study Group. Lancet 2000;356(9225):203–8.
- Rubio-Tapia A, Murray JA. Classification and management of refractory coeliac disease. Gut 2010;59(4):547–57.
- Rubio-Tapia A, Hill ID, Kelly CP, Calderwood AH, Murray JA. ACG clinical guidelines: diagnosis and management of celiac disease. Am J Gastroenterol 2013;108(5):656–76.





